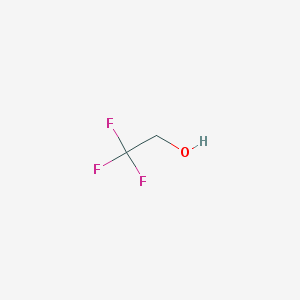
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
2020 is called a lost year due to the new crown epidemic, but it is of great significance for Chinese innovative drug companies.
2020 is called a lost year due to the new crown epidemic, but it is of great significance for Chinese innovative drug companies.
The number of domestic innovative drug companies authorized by foreign companies for original innovative drugs is more, and there are a total of 29 transactions from 2020 to February 2021.
Compared with domestic buyers, foreign pharmaceutical companies, especially multinational pharmaceutical giants, such as Novartis, AbbVie, Eli Lilly, Roche, Pfizer, etc.
Among them, BeiGene's PD-1 monoclonal antibody-Tilelizumab authorized Novartis with an advance payment of up to 650 million U.
Although China's innovative drug companies have continuously set new records for the external authorization of original innovative drugs, there is still a huge gap compared with the world's top innovative drug companies.
The total transaction price of the small molecule original innovative drug license-out announced amount is far less than that of the biological drug, which is only 13.
The external authorization of domestic innovative drug companies is dominated by emerging biotech companies, and the projects are mostly me-too, me-better, and first-in-class innovative drugs have not yet appeared, while the first-in-class international biotech companies in the same period have not yet appeared.
Bristol-Myers Squibb (BMS) announced in October 2020 that it would acquire MyoKardia, a small molecule innovative drug company focused on cardiomyopathy, for USD 13 billion.
The development of innovative drugs requires the accumulation of science and experience.