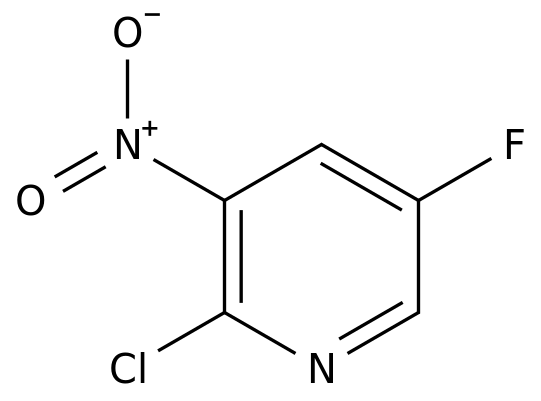
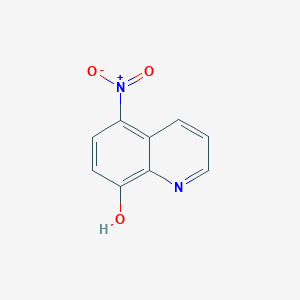
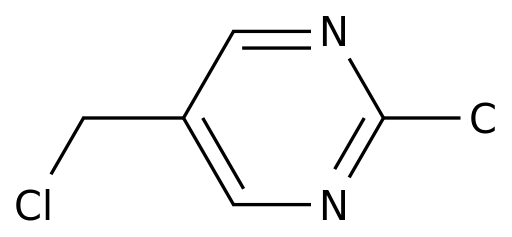
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
On September 16, the Jiangsu Provincial Drug Administration announced that, in accordance with the annual sampling work plan of the Jiangsu Provincial Drug Administration, the Jiangsu Provincial Drug Administration organized a sampling of drug production, operation and use units.
inspection found that a total of 11 batches of drugs did not meet the requirements, involving 10 varieties.
non-conforming items include chemical reactions, characteristics, sulfur dioxide residues, content determination, weight differences, etc.
11 batches of medicine, 2 batches were preparations, respectively, for injection of ammonia hydrochloride and proteasin anti-inflammatory tablets, and the rest were Chinese medicine tablets, involving sour kernels, light soybean meal, bone fat and so on.
present, for non-compliant drugs, the drug supervision and administration departments at all levels in Jiangsu Province have asked relevant enterprises and units to take risk control measures such as suspension of sales and use, to investigate the reasons for non-compliance and to carry out effective rectification.
drug supervision and administration departments at all levels in the province have organized and carried out investigation and punishment of relevant enterprises and units in accordance with the law.
: 12 batches of drugs are not qualified, all for drinking tablets on September 11, Gansu Provincial Drug Administration issued a drug quality announcement.
announced that by Jiayuguan City Food, Drug and Medical Device Inspection and Testing Center and other inspection and testing institutions, marked as Baoji Hanfang National Pharmaceutical Tablets Co., Ltd. and other 10 enterprises produced 12 batches of drugs do not meet the requirements.
It is worth noting that this sampling of unqualified drugs are Chinese medicine tablets, involving bran frying, gold and silver flowers, white fresh skin, grain fine grass, sea sand, small grass, Chaihu, white fresh skin, honey winter flowers, winter flowers, sheep and other varieties.
announcement pointed out that the relevant market supervision and administration departments have taken necessary control measures such as seizure and seizure for drugs that do not comply with the regulations, and have filed or are in the process of filing a case against the units involved in the product in accordance with relevant laws and regulations, and will publicly process the results within three months.
the Bureau requires that the municipal and state market supervision and administration bureaus strengthen supervision over unqualified products and related units involved, urge them to identify the causes of the problems, formulate and implement corrective measures, and effectively eliminate hidden risks.
: Traits refer to the appearance of the drug, texture, section, odor, taste, solubility, and physical constants.
drug identification tests are conducted on the basis of certain physical, chemical or biological characteristics of the drug, with the aim of identifying and studying the authenticity, safety and effectiveness of the drug.
content determination refers to the determination of the content of active ingredients in raw materials and preparations by chemical, instrument or biometric methods.
.