-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
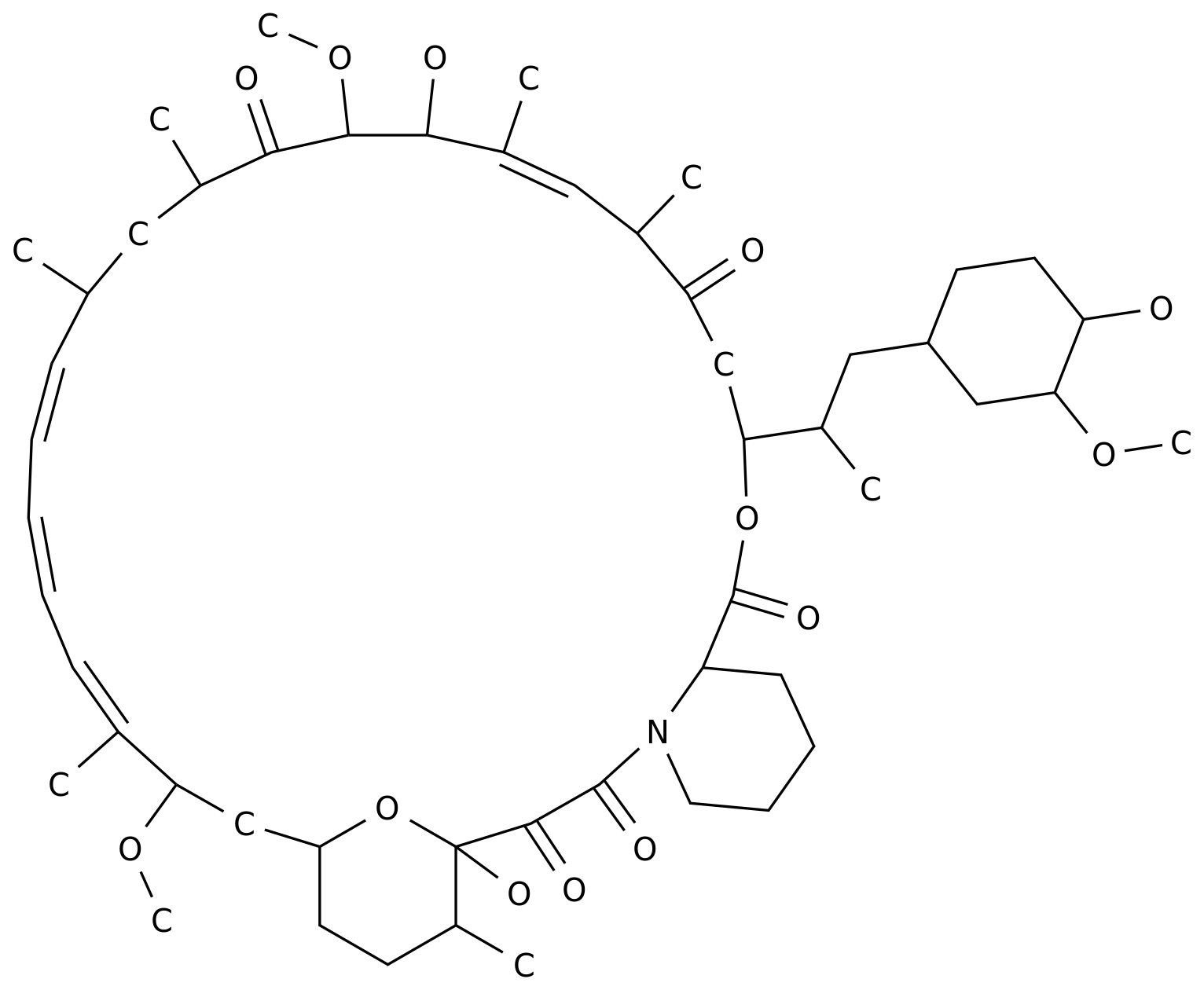
Upatinib is a Janus kinase inhibitor.
Upatinib is a Janus kinase inhibitor.
The study pooled treatment emergency adverse events (TEAEs) and laboratory data from five phase III trials.
The study pooled treatment emergency adverse events (TEAEs) and laboratory data from five phase III trials.
3834 patients received upatinib treatment, of which 15 mg (n = 2630) and 30 mg (n = 1204).
3834 patients received upatinib treatment, of which 15 mg (n = 2630) and 30 mg (n = 1204).
Compared with adalimumab, patients with upatinib have an increased risk of herpes zoster and CPK.
Compared with adalimumab, patients with upatinib have an increased risk of herpes zoster and CPK.
Original source:
Original source: Original source:Stanley B Cohen.
Stanley B Cohen.
bmj.
com/content/80/3/304" target="_blank" rel="noopener">Safety profile of upadacitinib in rheumatoid arthritis: integrated analysis from the SELECT phase III clinical programme.
Ann Rheum Dis.
February 2021.
bmj.
com/content/80/3/304" target="_blank" rel="noopener">Safety profile of upadacitinib in rheumatoid arthritis: integrated analysis from the SELECT phase III clinical programme.
Leave a message here