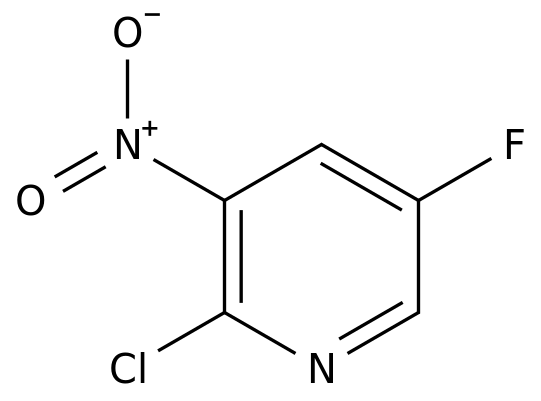
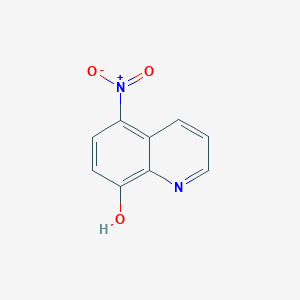
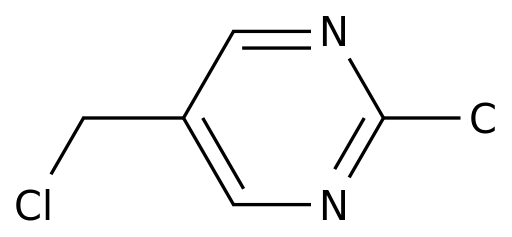
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
On June 1, the Beijing Municipal Drug Administration drafted the "Notice of the Beijing Municipal Drug Administration on Relevant Work Concerning the Reform of Drug Retail Business License Approval (Draft for Solicitation of Comments)" to solicit public opinions
.
The "Draft for Solicitation of Comments" clarifies that a new drug retail company should cancel the requirement of a feasible travel distance of more than 350 meters between an existing drug retail company and the new drug retail company or the original drug retail company that applies for a change of registered address , The distance to the existing pharmacy is no longer a condition for permit review
.
At the same time, the approval for the establishment of pharmaceutical retail companies was cancelled, and the original two approval processes of “preparation first” and “post-acceptance” were merged into one “new establishment” process, and the corresponding approval process was adjusted accordingly and the approval process was streamlined
.
It is reported that this is another super-first-tier city that has lifted distance restrictions on pharmacies after Shanghai this year
.
(Sina Pharmaceutical News)