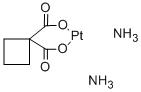
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
News April 5, 2021 //---For patients with advanced renal cell carcinoma (RCC), immune checkpoint blocking therapy can only control the condition of some patients, but the remaining patients are resistant The underlying mechanism is currently unclear.
In a study recently published in the journal Cancer Cell, Professor Eliezer M.
Van Allen from the Dana-Farber Cancer Institute and others treated cancer cells and immune cells before and after immune checkpoint blockade in patients with malignant RCC Transcriptomics analysis was performed at the single-cell level.
The results showed that some patients with effective treatments expressed higher levels of inhibitory receptor molecules in toxic T cells, and macrophages turned to pro-inflammatory characteristics after treatment.
In addition, there are two different subgroups of cancer cells, and their expression of angiogenic signals and immunosuppressive signals are significantly different.
Changes in the surface markers of cancer cells and up-regulation of immunosuppressive genes are accompanied by mutations in the PBRM1 gene.
These findings indicate that immune blocking therapy affects the microenvironment of RCC and affects the interaction between cancer cells and immune cells.
In a study recently published in the journal Cancer Cell, Professor Eliezer M.
Van Allen from the Dana-Farber Cancer Institute and others treated cancer cells and immune cells before and after immune checkpoint blockade in patients with malignant RCC Transcriptomics analysis was performed at the single-cell level.
The results showed that some patients with effective treatments expressed higher levels of inhibitory receptor molecules in toxic T cells, and macrophages turned to pro-inflammatory characteristics after treatment.
In addition, there are two different subgroups of cancer cells, and their expression of angiogenic signals and immunosuppressive signals are significantly different.
Changes in the surface markers of cancer cells and up-regulation of immunosuppressive genes are accompanied by mutations in the PBRM1 gene.
These findings indicate that immune blocking therapy affects the microenvironment of RCC and affects the interaction between cancer cells and immune cells.
(Image source: www.
cell.
com)
cell.
com)
First, the author collected tumor tissue samples from 8 patients, and 7 of them had already metastasized cancer cells.
On the day of collection, 5 patients received immune checkpoint blocker therapy, and 4 of them received both TKI therapy and immune checkpoint blockade therapy.
After that, the author performed single-cell sequencing and high-throughput exome sequencing on the collected cells.
On the day of collection, 5 patients received immune checkpoint blocker therapy, and 4 of them received both TKI therapy and immune checkpoint blockade therapy.
After that, the author performed single-cell sequencing and high-throughput exome sequencing on the collected cells.
All patients who received immune checkpoint blocking therapy received PD-1 targeting antibody therapy, so the authors believe that the therapy may affect the characteristics of immune cells in the patient's body.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
(Figure 1, Cell characteristics analysis of RCC patients before and after immune checkpoint blocking therapy)
Furthermore, the authors found that CD8 T cells can be divided into four different sets, one of which can cross the cell cycle, and the other highly expresses the MX1 gene downstream of interferon; the remaining two subgroups highly express TNFRSF9, which is responsible for the expression of one.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
(Figure 2, CD8 T cells "remodeled" after treatment in RCC patients)
In terms of macrophages, the authors found that after patients receive immune checkpoint blocking therapy, their macrophages will develop in the pro-inflammatory direction (ie M1), and the expression level of related pro-inflammatory genes will increase significantly.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
(Figure 3, tumor-associated macrophages undergo pro-inflammatory conversion after treatment in RCC patients)
In terms of cancer cells, the author has identified two different cancer cell subgroups, and their gene expression profiles are significantly different from each other.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
(Image source: www.
cell.
com)
cell.
com)
First, the author collected tumor tissue samples from 8 patients, and 7 of them had already metastasized cancer cells.
On the day of collection, 5 patients received immune checkpoint blocker therapy, and 4 of them received both TKI therapy and immune checkpoint blockade therapy.
After that, the author performed single-cell sequencing and high-throughput exome sequencing on the collected cells.
On the day of collection, 5 patients received immune checkpoint blocker therapy, and 4 of them received both TKI therapy and immune checkpoint blockade therapy.
After that, the author performed single-cell sequencing and high-throughput exome sequencing on the collected cells.
All patients who received immune checkpoint blocking therapy received PD-1 targeting antibody therapy, so the authors believe that the therapy may affect the characteristics of immune cells in the patient's body.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
(Figure 1, Cell characteristics analysis of RCC patients before and after immune checkpoint blocking therapy)
Furthermore, the authors found that CD8 T cells can be divided into four different sets, one of which can cross the cell cycle, and the other highly expresses the MX1 gene downstream of interferon; the remaining two subgroups highly express TNFRSF9, which is responsible for the expression of one.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
(Figure 2, CD8 T cells "remodeled" after treatment in RCC patients)
In terms of macrophages, the authors found that after patients receive immune checkpoint blocking therapy, their macrophages will develop in the pro-inflammatory direction (ie M1), and the expression level of related pro-inflammatory genes will increase significantly.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
(Figure 3, tumor-associated macrophages undergo pro-inflammatory conversion after treatment in RCC patients)
In terms of cancer cells, the author has identified two different cancer cell subgroups, and their gene expression profiles are significantly different from each other.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
First, the author collected tumor tissue samples from 8 patients, and 7 of them had already metastasized cancer cells.
On the day of collection, 5 patients received immune checkpoint blocker therapy, and 4 of them received both TKI therapy and immune checkpoint blockade therapy.
After that, the author performed single-cell sequencing and high-throughput exome sequencing on the collected cells.
On the day of collection, 5 patients received immune checkpoint blocker therapy, and 4 of them received both TKI therapy and immune checkpoint blockade therapy.
After that, the author performed single-cell sequencing and high-throughput exome sequencing on the collected cells.
All patients who received immune checkpoint blocking therapy received PD-1 targeting antibody therapy, so the authors believe that the therapy may affect the characteristics of immune cells in the patient's body.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
(Figure 1, Cell characteristics analysis of RCC patients before and after immune checkpoint blocking therapy)
Furthermore, the authors found that CD8 T cells can be divided into four different sets, one of which can cross the cell cycle, and the other highly expresses the MX1 gene downstream of interferon; the remaining two subgroups highly express TNFRSF9, which is responsible for the expression of one.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
(Figure 2, CD8 T cells "remodeled" after treatment in RCC patients)
In terms of macrophages, the authors found that after patients receive immune checkpoint blocking therapy, their macrophages will develop in the pro-inflammatory direction (ie M1), and the expression level of related pro-inflammatory genes will increase significantly.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
(Figure 3, tumor-associated macrophages undergo pro-inflammatory conversion after treatment in RCC patients)
In terms of cancer cells, the author has identified two different cancer cell subgroups, and their gene expression profiles are significantly different from each other.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
All patients who received immune checkpoint blocking therapy received PD-1 targeting antibody therapy, so the authors believe that the therapy may affect the characteristics of immune cells in the patient's body.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
In order to verify this conjecture, the author performed a cluster analysis of lymphocytes and obtained 12 different subgroups, including B cells, plasma cells, NK cells, NKT cells, regulatory T cells, memory and effector helper T cells.
Cells and so on.
(Figure 1, Cell characteristics analysis of RCC patients before and after immune checkpoint blocking therapy)
Furthermore, the authors found that CD8 T cells can be divided into four different sets, one of which can cross the cell cycle, and the other highly expresses the MX1 gene downstream of interferon; the remaining two subgroups highly express TNFRSF9, which is responsible for the expression of one.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
(Figure 2, CD8 T cells "remodeled" after treatment in RCC patients)
In terms of macrophages, the authors found that after patients receive immune checkpoint blocking therapy, their macrophages will develop in the pro-inflammatory direction (ie M1), and the expression level of related pro-inflammatory genes will increase significantly.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
(Figure 3, tumor-associated macrophages undergo pro-inflammatory conversion after treatment in RCC patients)
In terms of cancer cells, the author has identified two different cancer cell subgroups, and their gene expression profiles are significantly different from each other.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
Furthermore, the authors found that CD8 T cells can be divided into four different sets, one of which can cross the cell cycle, and the other highly expresses the MX1 gene downstream of interferon; the remaining two subgroups highly express TNFRSF9, which is responsible for the expression of one.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
A costimulatory factor called 4-1BB.
In addition, PDCD1 (encoding PD-1), HAVCR2 (encoding TIM-3) and LAG3 are also significantly expressed, and the expression levels of these genes are more significant in cells with high 4-1BB expression.
On this basis, the authors found that 4-1BB-Lo CD8+ T cells showed up-regulation of genes such as GZMA and FASLG after the patient received immune checkpoint blocking therapy, which means that these cells have the potential to differentiate in the mid-terminal direction.
(Figure 2, CD8 T cells "remodeled" after treatment in RCC patients)
In terms of macrophages, the authors found that after patients receive immune checkpoint blocking therapy, their macrophages will develop in the pro-inflammatory direction (ie M1), and the expression level of related pro-inflammatory genes will increase significantly.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
(Figure 3, tumor-associated macrophages undergo pro-inflammatory conversion after treatment in RCC patients)
In terms of cancer cells, the author has identified two different cancer cell subgroups, and their gene expression profiles are significantly different from each other.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
In terms of macrophages, the authors found that after patients receive immune checkpoint blocking therapy, their macrophages will develop in the pro-inflammatory direction (ie M1), and the expression level of related pro-inflammatory genes will increase significantly.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
On the other hand, the expression levels of genes such as VSIR, VSIG4, PD-L2 and SIGLEC10 in tumor-associated macrophages in patients who responded to treatment were significantly higher than those in patients who did not respond.
These results indicate that it also has the property of inhibiting inflammatory response.
(Figure 3, tumor-associated macrophages undergo pro-inflammatory conversion after treatment in RCC patients)
In terms of cancer cells, the author has identified two different cancer cell subgroups, and their gene expression profiles are significantly different from each other.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
In terms of cancer cells, the author has identified two different cancer cell subgroups, and their gene expression profiles are significantly different from each other.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
Among them, TP1 cells have significantly higher expression of genes related to cell adhesion and angiogenesis, while TP2 cells have higher expression levels of glycolysis and oxidative phosphorylation genes.
Furthermore, the authors found that after receiving immune blocking therapy, nectin-2 in TP1 cells was significantly upregulated, while the expression of several immunosuppressive genes in TP2 cells was significantly increased.
These changes will make cancer cells more likely to escape immune cells.
Of killing.
Finally, the authors found that there is a significant correlation between TP1-related gene expression and patient survival after treatment.
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
(Figure 4, Classification and characteristic analysis of cancer cells in RCC patients after treatment)
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
In summary, the author analyzed the gene expression of different cell types in RCC patients after receiving immuno-blocking therapy through systematic single-cell transcriptomics methods.
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
The above results provide future RCC-related research and the development of clinical treatment methods.
New ideas.
(Bioon.
com)
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
Original source: Kevin Bi, Meng Xiao He, Z iad Bakouny et al.
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
Original source: iad Bakouny et al. , Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.
(2021) DOI: https://doi.
org/10.
1016/j.
ccell .
2021.
02.
015
, Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma.
Cancer Cell.