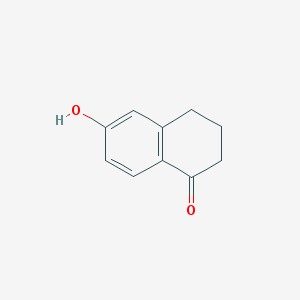
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
According to the news on June 29, the National Medical Products Administration of the State Drug Administration of the State Drug Administration of the Quality and Efficacy Consistency Evaluation Expert Committee confirmed that the list of generic reference preparations (the 42nd batch) is now released
.
Hereby inform
.
Attachment: List of Generic Drug Reference Preparations (Forty-second batch)
National Food and Drug Administration June 22, 2021
.
Hereby inform
.
Attachment: List of Generic Drug Reference Preparations (Forty-second batch)
| Serial number | Generic name of the drug | English name/trade name | Specification | Licensee | Remark 1 | Remark 2 |
|---|---|---|---|---|---|---|
| | Ezetimibe Simvastatin Tablets | Ezetimibe and Simvastatin Tablets/Bao Zhineng | Each tablet contains 10mg of ezetimibe and 20mg of simvastatin | Merck Sharp & Dohme BV | Original drugs marketed in China | Original research import |
| | Ezetimibe Simvastatin Tablets | Ezetimibe and Simvastatin Tablets/Bao Zhineng | Each tablet contains 10mg of ezetimibe and 40mg of simvastatin | Merck Sharp & Dohme BV | Original drugs marketed in China | Original research import |
| | Granisetron transdermal patch | Granisetron Transdermal Patches | 34. 3mg/52cm2 (released amount 3. 1mg/24h) | ProStrakan | Original drugs marketed in China | Original research import |
| | Omega-3 fish oil fat emulsion injection | ω-3 Fish Oil Fat Emulsion Injection/Omegaven | 50ml: 5g (refined fish oil): 0. 6g (lecithin) | Fresenius Kabi Austria GmbH | Original drugs marketed in China | Original research import |
| | Obucaine Hydrochloride Eye Drops | Oxybuprocaine Hydrochloride Eye Drops / Benoxil | 0. 4% (0. 5ml:2mg) | Santen Pharmaceutical Co. , Ltd. | Original drugs marketed in China | Original research import |
| | 10% fat emulsion (OO)/5. 5% amino acid (15)/glucose (20%) injection | 10% Fat Emulsion(OO)/5. 5% Amino Acids(15)/Glucose(20%) Injection/Oliclinomel | 1500ml: 10% olive oil fat emulsion 300ml+5. 5% compound amino acid injection (15) 600ml+20% glucose injection 600ml | Baxter SA | Original drugs marketed in China | Original research import |
| | 10% fat emulsion (OO)/5. 5% amino acid (15)/glucose (20%) injection | 10% Fat Emulsion(OO)/5. 5% Amino Acids(15)/Glucose(20%) Injection/Oliclinomel | 1000ml: 10% olive oil fat emulsion 200ml+5. 5% compound amino acid injection (15) 400ml+20% glucose injection 400ml | Baxter SA | Original drugs marketed in China | Original research import |
| | Sodium Benzoate Sodium Phenylacetate Injection | Sodium Phenylacetate and Sodium Benzoate Injection /Ammonul | 10%; 10% (5g/50mL; 5g/50mL) | Bausch Health Us LLC | No imported original research drugs | American Orange Book |
| | Glyceryl Phenylbutyrate Oral Liquid | Glycerol Phenylbutyrate Oral Liquid/Ravicti | 1. 1 g/mL | Horizon Therapeutics LLC | No imported original research drugs | American Orange Book |
| | Cetirizine Hydrochloride Injection | Cetirizine Hydrochloride Injection/Quzyttir | 10mg/ml | JDP Therapeutics LLC | No imported original research drugs | American Orange Book |
| | Lamotrigine sustained-release tablets | Lamotrigine Extended Release Tablets / Lamictal XR | 25mg | GlaxoSmithKline LLC | No imported original research drugs | American Orange Book |
| | Lamotrigine sustained-release tablets | Lamotrigine Extended Release Tablets / Lamictal XR | 50mg | GlaxoSmithKline LLC | No imported original research drugs | American Orange Book |
| | Lamotrigine sustained-release tablets | Lamotrigine Extended Release Tablets / Lamictal XR | 100mg | GlaxoSmithKline LLC | No imported original research drugs | American Orange Book |
| | Lamotrigine sustained-release tablets | Lamotrigine Extended Release Tablets / Lamictal XR | 200mg | GlaxoSmithKline LLC | No imported original research drugs | American Orange Book |
| | Lamotrigine sustained-release tablets | Lamotrigine Extended Release Tablets / Lamictal XR | 250mg | GlaxoSmithKline LLC | No imported original research drugs | American Orange Book |
| | Lamotrigine sustained-release tablets | Lamotrigine Extended Release Tablets / Lamictal XR | 300mg | GlaxoSmithKline LLC | No imported original research drugs | American Orange Book |
| | Bupropion Hydrobromide Sustained Release Tablets | Bupropion Hydrobromide Extended-Release Tablets/Aplenzin | 174mg | Valeant Pharmaceuticals North America LLC | No imported original research drugs | American Orange Book |
| | Bupropion Hydrobromide Sustained Release Tablets | Bupropion Hydrobromide Extended-Release Tablets/Aplenzin | 348mg | Valeant Pharmaceuticals North America LLC | No imported original research drugs | American Orange Book |
| | Bupropion Hydrobromide Sustained Release Tablets | Bupropion Hydrobromide Extended-Release Tablets/Aplenzin | 522mg | Valeant Pharmaceuticals North America LLC | No imported original research drugs | American Orange Book |
| | Riboflavin sodium phosphate eye drops | Riboflavin 5'-Phosphate Ophthalmic Solution/ Photrexa | 0. 146% | Glaukos Corporation | No imported original research drugs | American Orange Book |
| | Azelaic Acid Gel | Azelaic Acid Gel / Finacea | 15% | Leo Pharma AS | No imported original research drugs | American Orange Book |
| | Gemcitabine Hydrochloride Injection | Gemcitabine Hydrochloride Injection | 200mg/5. 26ml (38 mg/ml) | Hospira, Inc. | No imported original research drugs | American Orange Book |
| | Gemcitabine Hydrochloride Injection | Gemcitabine Hydrochloride Injection | 1g/26. 3ml (38 mg/ml) | Hospira, Inc. | No imported original research drugs | American Orange Book |
| | Idarubicin Hydrochloride Injection | Idarubicin Hydrochloride Injection/Idamycin Pfs | 1 mg/ml | Pfizer Inc | Unimported original research | American Orange Book |
| | Fluoxetine Hydrochloride Oral Solution | Fluoxetine Hydrochloride Oral Solution/ Prozac | 20mg/5ml | Lilly France | No imported original research drugs | EU listing |
| | Triamcinolone acetonide and econazole cream | Triamcinolone Acetonide and Econazole Nitrate Cream | Each gram contains econazole nitrate 10mg, triamcinolone acetonide 1. 0mg | Janssen-Cilag | No imported original research drugs | EU listing |
| | Terbinafine Gel | Terbinafine Gel/Lamisil DermGel | 1% | Glaxosmithkline Sante Grand Public | No imported original research drugs | EU listing |
| | ω-3 fish oil medium and long-chain fat emulsion/amino acid (16)/glucose (16%) injection | ω-3 Fish Oil Medium and Long Chain Fat Emulsion/Amino Acids (16)/Glucose (16%) Injection / NuTRIflex Omega peri | 1250ml | B. Braun Melsungen AG | No imported original research drugs | EU listing |
| | Compound alpha-keto acid tablets | Compound α-Ketoacid Tablets | 0. 63g | Fresenius Kabi Deutschland GmbH | No imported original research drugs | EU listing |
| | Cefcapene Pivoxil Hydrochloride Granules | Cefcapene Pivoxil Hydrochloride Granules /Flomox | 10% (1g 100mg containing hydrochloric acid salt hydrate, 50mg/bag) | Shiono Yoshihide Pharmaceutical Co. , Ltd. | No imported original research drugs | Listed in Japan |
| | Melatonin granules for children | Melatonin granules for pediatric / Melatobel | 1g: 2mg | ノーベルファーマ Co. , Ltd. | No imported original research drugs | Listed in Japan |
| | Nificaran Hydrochloride for Injection | Nifekalant Hydrochloride for Injection / Shinbit | 50mg | トアエイヨー Co. , Ltd. | No imported original research drugs | Listed in Japan |
| | Iron Citrate Tablets | Ferric Citrate Tablet/ Riona Tab 250mg | 250mg (calculated as iron citrate) | Torii Pharmaceutical Co. , Ltd. | No imported original research drugs | Listed in Japan |
| | Baclofen Oral Solution | Baclofen Oral Solution/Lioresal | 5mg/5ml | Novartis Pharmaceuticals UK Limited | No imported original research drugs | UK listed |
| | Atenolol injection | Atenolol Injection/Tenormin | 10ml:5mg | AstraZeneca | No imported original research drugs | UK listed |
| | Paroxetine Hydrochloride Tablets | Paroxetine Hydrochloride Tablets/Sailote | 20mg (calculated as C19H20FNO3) | Sino-U. S. Tianjin SmithKline Pharmaceutical Co. , Ltd. | Drugs produced in China by foreign original research companies that have been reviewed and determined | Original research real estate |
| | Salbutamol Sulfate Inhalation Aerosol | Salbutamol Sulfate Inhalation Aerosol/Proventil-Hfa | 90μg/press (calculated as salbutamol) | Kindeva Drug Delivery LP/Merck Sharp & Dohme Corp. /3M Drug Delivery Systems | No imported original research drugs | American Orange Book |
| | Pantoprazole Sodium for Injection | Pantoprazole Sodium For Injection/ Pantoprazole Sodium | 40mg | Hikma Pharmaceuticals LLC | No imported original research drugs | American Orange Book |
| | Allopurinol Tablets | Allopurinol Tablets /Zyloprim | 0. 3g | Casper Parma LLC | No imported original research drugs | American Orange Book |
| | Enalapril Maleate Tablets | Enalapril Maleate Tablets/Vasotec | 5mg | Bausch Health US LLC | No imported original research drugs | American Orange Book |
| | Enalapril Maleate Tablets | Enalapril Maleate Tablets/Vasotec | 10mg | Bausch Health US LLC | No imported original research drugs | American Orange Book |
| | Enalapril Maleate Tablets | Enalapril Maleate Tablets/Vasotec | 20mg | Bausch Health US LLC | No imported original research drugs | American Orange Book |
| | Cabazitaxel injection | Cabazitaxel Injection/Jevtana | 60mg/1. 5ml (40mg/ml) | Sanofi-Aventis US INC | No imported original research drugs | American Orange Book |
| | Olapali tablets | Olaparib Tablets/Lynparza | 100mg | AstraZeneca Pharmaceuticals LP | No imported original research drugs | American Orange Book |
| | Olapali tablets | Olaparib Tablets/Lynparza | 150mg | AstraZeneca Pharmaceuticals LP | No imported original research drugs | American Orange Book |
| | Trifluridine Tepipyrimidine Tablets | Trifluridine and Tipiracil Hydrochloride Tablets/ Lonsurf | Trifluridine 15mg, tepipyridine hydrochloride 7. 065mg (equivalent to tepipyridine 6. 14mg) | Taiho oncology Inc. | No imported original research drugs | American Orange Book |
| | Trifluridine Tepipyrimidine Tablets | Trifluridine and Tipiracil Hydrochloride Tablets/ Lonsurf | Trifluridine 20mg, tepipyridine hydrochloride 9. 420mg (equivalent to tepipyridine 8. 19mg) | Taiho oncology Inc | No imported original research drugs | American Orange Book |
| | Paroxetine Hydrochloride Tablets | Paroxetine Hydrochloride Tablets / Paxil | 10mg | Apotex Technologies Inc | No imported original research drugs | American Orange Book |
| | Paroxetine Hydrochloride Tablets | Paroxetine Hydrochloride Tablets / Paxil | 30mg | Apotex Technologies Inc | No imported original research drugs | American Orange Book |
| | Brivaracetam injection | Brivaracetam Solution for Injection/Briviact | 10mg/ml | UCB Inc | No imported original research drugs | American Orange Book |
| | Fluticasone Propionate Cream | Fluticasone Propionate Cream | 0. 05% | Perrigo Israel Pharmaceuticals Ltd | Internationally recognized drugs of the same kind | American Orange Book |
| | Latanoprost eye drops | Latanoprost Eye Drops/Xalatan | 0. 005% | Upjohn US 2 LLC | No imported original research drugs | American Orange Book |
| | Carprost tromethamine injection | Carboprost Tromethamine Injection/Hemabate | 1ml: 250μg | Pfizer Inc | No imported original research drugs | American Orange Book |
| | Tofacitib Citrate Sustained Release Tablets | Tofacitinib Citrate Extended Release tablets/Xeljanz Xr | 22mg | Pfizer Inc | No imported original research drugs | American Orange Book |
| | Cefoperazone Sodium and Sulbactam Sodium for Injection | Cefoperazone Sodium and Sulbactam Sodium for Injection/Sulperazon | 1g (Cefoperazone 0. 5g: Sulbactam 0. 5g) | Pfizer Polska Sp. z oo | No imported original research drugs | EU listing |
| | Etibatide Injection | Eptifibatide Injection/Integrilin | 20mg/10mL | GlaxoSmithKline (Ireland) Limited | No imported original research drugs | EU listing |
| | Etibatide Injection | Eptifibatide Injection/Integrilin | 75mg/100mL | GlaxoSmithKline (Ireland) Limited | No imported original research drugs | EU listing |
| | Rivaroxaban tablets | Rivaroxaban Tablets/Xarelto | 2. 5mg | Bayer AG | No imported original research drugs | EU listing |
| | Rivaroxaban tablets | Rivaroxaban Tablets/Xarelto | 10mg | Bayer AG | No imported original research drugs | EU listing |
| | Rivaroxaban tablets | Rivaroxaban Tablets/Xarelto | 15mg | Bayer AG | No imported original research drugs | EU listing |
| | Rivaroxaban tablets | Rivaroxaban Tablets/Xarelto | 20mg | Bayer AG | No imported original research drugs | EU listing |
| | Gadoterate meglumine injection | Gadoteric Acid Meglumine Salt Injection/Dotarem | 0. 5 mmol/mL (10ml, 15ml, 20ml/bottle) | Guerbet | No imported original research drugs | EU listing |
| | Vardenafil Hydrochloride Orally Disintegrating Tablets | Vardenafil Hydrochloride Orally disintegrating Tablets/Levitra | 10mg | Bayer AG | No imported original research drugs | EU listing |
| | Methylprednisolone tablets | Methylprednisolon /Medrol | 16mg | Pfizer Italia srl | No imported original research drugs | EU listing |
| | Carrosol Sodium Injection | Carbosulfan Sodium injection/Adona | 20ml: 100mg | Nipro ES Fame Co. , Ltd. | No imported original research drugs | Listed in Japan |
| | Torsemide tablets | Torasemide Tablets/Torem | 5mg | Mylan Products Ltd. | No imported original research drugs | UK listed |
| | Torsemide tablets | Torasemide Tablets/Torem | 10mg | Mylan Products Ltd. | No imported original research drugs | UK listed |
| 6-12 | Hydroxychloroquine Sulfate Tablets | Hydroxychloroquine Sulfate Tablets/Plaquenil | 0. 2g | Aventis Pharma Limited/ Sanofi-aventis Ireland Ltd. T/A SANOFI | | ,Sanofi-aventis Ireland Ltd. T/A SANOFI |
| 10-223 | | Crizotinib Capsules/XALKORI | 0. 25g | Pfizer Ltd. /PFIZER EUROPE MA EEIG | | ,PFIZER EUROPE MA EEIG |
| 10-224 | | Crizotinib Capsules/XALKORI | 0. 2g | Pfizer Ltd. /PFIZER EUROPE MA EEIG | | ,PFIZER EUROPE MA EEIG |
| 17-7 | | Benidipine Hydrochloride Tablets/ | 4mg | /Kyowa Kirin Co. , Ltd. | | ,Kyowa Kirin Co. , Ltd. |
| 17-8 | | Benidipine Hydrochloride Tablets/ | 8mg | /Kyowa Kirin Co. , Ltd. | | ,Kyowa Kirin Co. , Ltd. |
| 17-15 | | Cinacalcet Hydrochloride Tablets/ | 25mg | /Kyowa Kirin Co. , Ltd. | | ,Kyowa Kirin Co. , Ltd. |
| 17-16 | | Cinacalcet Hydrochloride Tablets/ | 75mg | /Kyowa Kirin Co. , Ltd. | | ,Kyowa Kirin Co. , Ltd. |
| 23-18 | | Lenvatinib Mesilate Capsules/Lenvima() | 4mg | Eisai Europe Ltd. / Eisai GmbH | | ,Eisai GmbH |
| 24-179 | | Tafluprost Eye Drops/Tapros() | 0. 0015% (2. 5ml:37. 5μg) | Santen Pharmaceutical Co. ,Ltd. | Original drugs marketed in China | The specification is updated to 0. 0015% (2. 5ml:37. 5μg) |
| 27-235 | Beraprost Sodium Tablets | Beraprost Sodium Tablets | 40μg | Takeda Pharma Japan Inc. (Teva Pharma Japan Inc) | Internationally recognized drugs of the same kind | Specification revised to 40μg |
| Remarks | 1. The generic names, dosage forms, etc. of the varieties listed in the catalog that have not yet been marketed in China shall be subject to the approval of the Pharmacopoeia Commission . 2. After the publication of the reference preparation catalogue, the varieties that have not been officially released will be subject to special research and will be released separately according to the research results . 3. The reference preparations marketed in the EU include the same drugs marketed in the UK . 4. The selection of unimported reference preparations to carry out generic drug research not only meets its quality requirements, but also needs to meet the requirements of the current version of the Chinese Pharmacopoeia and related guidelines . |
National Food and Drug Administration June 22, 2021