-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
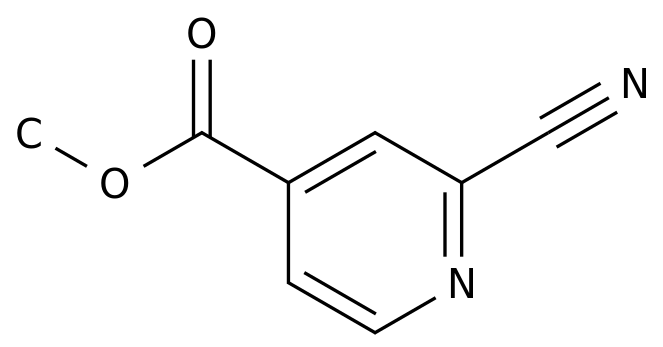
On October 30, the official website of the State Drug Administration showed that Dongsan pharmaceutical products to imitate the 4 categories of reported Tegrelo tablets into the administrative approval stage, is expected to be approved in recent days and treated as a review.
is an anti-thrombosis drug, Mienet data show that in 2019 China's public medical institutions terminal Grelo sales of more than 1.5 billion yuan, 2020H1 sales of more than 900 million yuan.
is a new anti-thrombosis drug, a direct-acting, reversible combination of P2Y12 plateplate inhibitors.
does not need to be metabolized and active in the liver, and has the characteristics of fast and powerful.
, which was originally researched by AstraZeneta, is called Perinda, which has global sales of $1,581 million in 2019. according to
meters of intranet data, in 2019 China's urban public hospitals, county-level public hospitals, urban community centers and township hospitals (China's public medical institutions) terminal sales of more than 1.5 billion yuan, an increase of 48.85 percent year-on-year;
China's public medical institutions terminal for Grelo sales source: Minet China's public medical institutions terminal competition pattern in the current domestic market for Grelo products have for Grelo tablets, for Grelo dispersal film.
In addition to the original research astraZeneca, there are eight generic pharmaceutical companies such as Xinlitai, Zhengda Tianqing, Shi Pharmaceutical Group, Yangzijiang, etc.
, although It was included in the third batch, it was hard to resist the enthusiasm of drug companies for imitation.
meters of intranet data show that Chengdu Beit, Hesco, Lepu Pharmaceuticals, Luoxin Pharmaceuticals, Colum Pharmaceuticals and other 19 pharmaceutical companies will be modeled after 4 categories of reported Tegrelo tablets in the review.
the registration status of The Grelo tablets of Sundance Drug has been changed to "in approval" and is expected to be approved and evaluated in the near past.
source: MiNet Database, NMPA