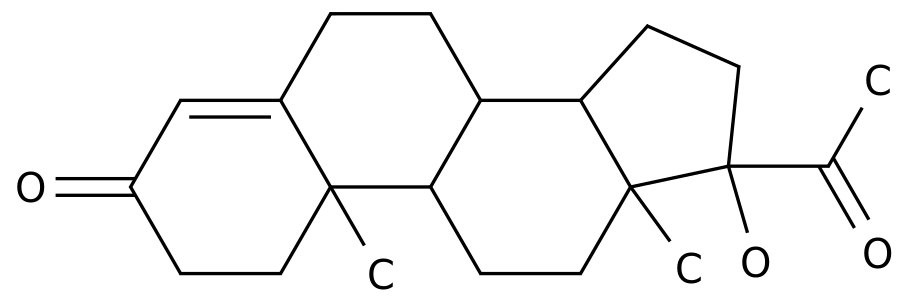
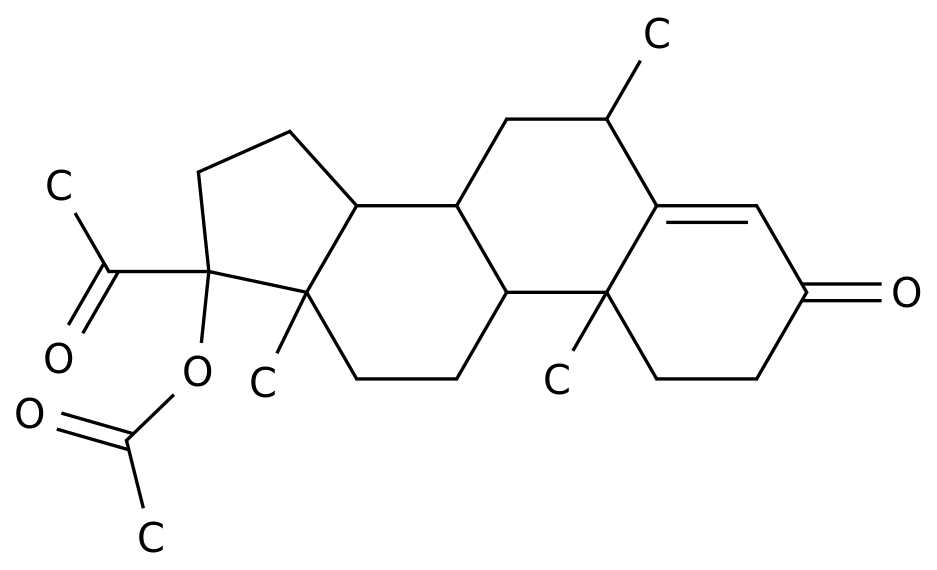
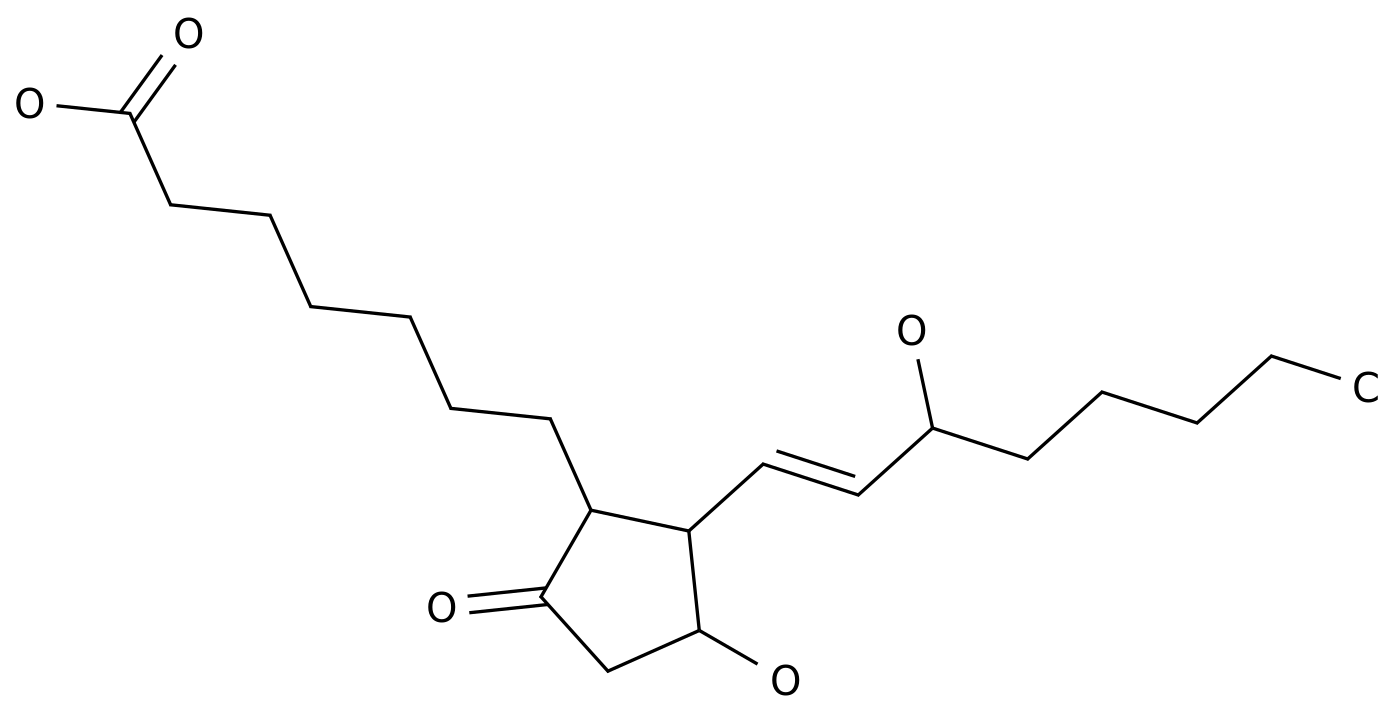
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
In the past 20 years, the global prevalence of obesity in children and adolescents has increased significantly, and there are now more than 124 million children and adolescents suffering from obesity in the world.
Childhood diabetes
Pharmaceutical company Novo Nordisk (Novo Nordisk) today announced that the European Medicines Management Agency (EMA) who proposes to extend the range of indications with drugs Saxenda Commission (CHMP), for the treatment of obesity in young people aged 12-17.
management
If approved, Saxenda will become the first adolescent obesity treatment drug approved by the European Union.
2 2 2
CHMP’s positive opinion is based on the results of a phase III trial published last year in the New England Journal of Medicine.
When Saxenda is used as an adjunct to lifestyle therapy, the body mass index (BMI) and weight of obese adolescents are significantly reduced.
Original source:
Original source:href="" target="_blank" rel="noopener">