-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
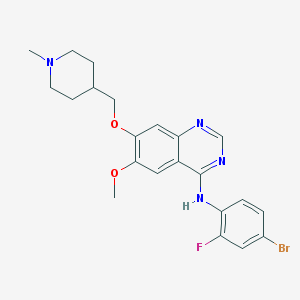
▎Editor of WuXi AppTec's content team On September 28, 2021, Artios Pharma announced that its highly selective DNA polymerase θ (Pol θ) inhibitor ART4215 has been administered to the first patient in a phase 1/2a clinical trial
.
The press release states that this is the first oral specific small molecule Polθ inhibitor to enter clinical development
.
This open-label, multi-center clinical trial will evaluate the safety, tolerability, pharmacokinetics and clinical effects of oral ART4215 as a single agent or in combination with other anti-cancer drugs in patients with advanced or metastatic solid tumors It is planned to enroll 206 patients, and it is expected that mid-term safety and tolerability data will be obtained in 2022
.
Artios is actively developing a promising product line of potential "best-in-class" and "first-in-class" DNA damage response (DDR) therapy, which can inhibit new types of DNA repair in tumors with weakened DNA damage response.
Target, such as Polθ
.
These new therapies will use certain genetic defects in the DNA repair process to selectively destroy cancer cells without harming normal cells
.
Image source: 123RF is a DNA polymerase, Polθ is involved in repairing DNA double-strand breaks
.
It is overexpressed in many tumors and is expressed at low levels in healthy tissues
.
Studies have shown that, as a single agent, ART4215 has anti-cancer activity in patients who have progressed after PARP inhibitor treatment.
It can also be used in combination with PARP inhibitors to treat patients who have not received PARP inhibitor treatment, or with other DNA damage therapies ( Such as ionizing radiation and cytotoxic chemotherapy) combined use
.
The principal investigator of the clinical trial, Dr.
Erika P.
Hamilton, said: “The preclinical data of ART4215 is encouraging and it has the potential to overcome resistance to PARP inhibitors and other DNA damage therapies
.
We look forward to providing more effective treatment options for patients.
Promote the progress of this class of new inhibitors
.
"Reference: [1] Artios Doses First Patient in Phase 1/2a Study of Polθ Inhibitor ART4215.
Retrieved September 28, 2021, from https:// /09/28/artios-doses-first-patient-in-phase-1-2a-study-of-pol%ce%b8-inhibitor-art4215/Disclaimer: WuXi AppTec's content team focuses on introducing global biomedical health research Progress
.
This article is for the purpose of information exchange only.
The opinions expressed in the article do not represent the position of WuXi AppTec, nor does it mean that WuXi AppTec supports or opposes the views in the article
.
This article is not a treatment recommendation either
.
If you need guidance on the treatment plan, please go to a regular hospital for treatment
.
.
The press release states that this is the first oral specific small molecule Polθ inhibitor to enter clinical development
.
This open-label, multi-center clinical trial will evaluate the safety, tolerability, pharmacokinetics and clinical effects of oral ART4215 as a single agent or in combination with other anti-cancer drugs in patients with advanced or metastatic solid tumors It is planned to enroll 206 patients, and it is expected that mid-term safety and tolerability data will be obtained in 2022
.
Artios is actively developing a promising product line of potential "best-in-class" and "first-in-class" DNA damage response (DDR) therapy, which can inhibit new types of DNA repair in tumors with weakened DNA damage response.
Target, such as Polθ
.
These new therapies will use certain genetic defects in the DNA repair process to selectively destroy cancer cells without harming normal cells
.
Image source: 123RF is a DNA polymerase, Polθ is involved in repairing DNA double-strand breaks
.
It is overexpressed in many tumors and is expressed at low levels in healthy tissues
.
Studies have shown that, as a single agent, ART4215 has anti-cancer activity in patients who have progressed after PARP inhibitor treatment.
It can also be used in combination with PARP inhibitors to treat patients who have not received PARP inhibitor treatment, or with other DNA damage therapies ( Such as ionizing radiation and cytotoxic chemotherapy) combined use
.
The principal investigator of the clinical trial, Dr.
Erika P.
Hamilton, said: “The preclinical data of ART4215 is encouraging and it has the potential to overcome resistance to PARP inhibitors and other DNA damage therapies
.
We look forward to providing more effective treatment options for patients.
Promote the progress of this class of new inhibitors
.
"Reference: [1] Artios Doses First Patient in Phase 1/2a Study of Polθ Inhibitor ART4215.
Retrieved September 28, 2021, from https:// /09/28/artios-doses-first-patient-in-phase-1-2a-study-of-pol%ce%b8-inhibitor-art4215/Disclaimer: WuXi AppTec's content team focuses on introducing global biomedical health research Progress
.
This article is for the purpose of information exchange only.
The opinions expressed in the article do not represent the position of WuXi AppTec, nor does it mean that WuXi AppTec supports or opposes the views in the article
.
This article is not a treatment recommendation either
.
If you need guidance on the treatment plan, please go to a regular hospital for treatment
.