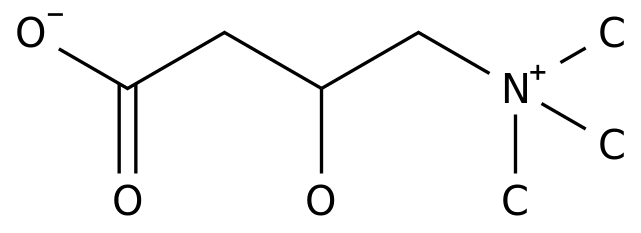
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Targetpregnant women with inflammatory bowel disease (IBD) have an early discontinuation of infrasix (IFX) that reduces the risk of exposure to the drug in the fetus, but may increase the risk of onset of the disease, leading to poor pregnancy outcomesThe effects of early IFX discontinuation on maternal disease activity and their high-risk infants were assessed in this studymethod sed
methods In a retrospective study of the Truven Health Analytics MarketScan database between 2011 and 2015, the researchers compared the clinical outcomes of IBD patients who interrupted IFX for more than 90 days ("early IFX") before childbirth with those who interrupted IFX for less than 90 days (or less)results
After IFX is discontinued, the early IFX group (68 deliveries) needed more steroid prescriptions to control disease activity (P.001) than the late IFX group (318 births)The early IFX group had more premature babies (P.049), but there was no difference in intrauterine developmental delays, pregnancy rates and stillbirth rates between the two groupsSimilarly, acute respiratoryinfections in infants of infants in the late IFX group with early IFX group, and developmental delays and congenital malformations did not increaseconclusions
When pregnant women stop IFX early in pregnancy, mothers without steroid snobs are at risk of developing the disease and preterm birthAt least in the first year of life, it seems safe to continue using IFX