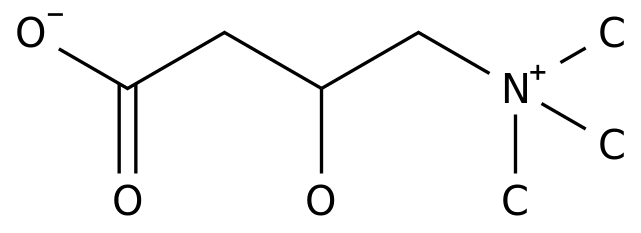
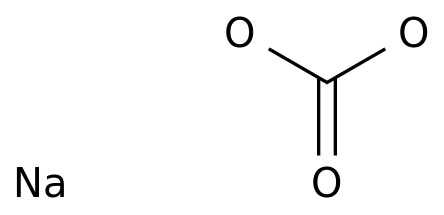
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Crohn's disease (CD) and ulcerative colitis (UC) are part of a group of idepathic autoimmune inflammatory bowel disease (IBDs) characterized by the subsequent remission of the disease.
usually involves mucous membrane inflammation, which eventually leads to changes in the structure and function of the upper intestinal cortitis.
, the main goal of IBD therapy is to induce and sustain clinical remission.
nevertheless, even in the case of effective treatment, subclinical inflammation can persist, leading to an increased risk of recurrence and an increased risk of upgrading treatment to immunomodulants, biotherapy and even surgery.
biopsy samples collected during endoscopy are still the gold standard for assessing intestinal inflammation, but they are invasive and can lead to misleading results if the endoscope fails to reach the lesions.
, biomarkers have become important indicators of disease activity, risk of recurrence, increased treatment needs and/or response to treatment.
serum dispeptidease 4 (DPP-4) as a biomarker for inflammatory bowel disease (IBD) has attracted the interest of researchers because it can instain several peptides involved in inflammatory cascading reactions.
, this study explores this.
researchers used clinical indicators to evaluate two forward-looking queues of 195 patients (101 with Crohn's disease (CD) and 94 with ulcerative colitis (UC)) and follow-up to assess the effectiveness of the treatment.
68 patients underwent endoscopy at the baseline.
the treatment response was evaluated in a second group of 46 biologically treated patients.
quantitative analysis of serum DEP-4, C-reactive protein (CRP) and fecal calcium-guard protein levels over baseline and follow-up.
results showed a significant decrease in median DPP-4 levels in active IBD patients compared to those in remission (CD:1043 (831-1412) vs 1589 (1255-1956) ng / mL;P slt;0.001; UC:1317 (1058-1718) vs 1798 (1329-2305) ng / mL; P - 0.001) and health control group (2175 ( 1875-3371) ng / mL).
fact, DPP-4 distinguishes between clinical and endoscopic activity and mitigation, with a sub-curve area (AUC) of 0.81 / 0.93 (CD) and 0.71 / 0.79 (UC), in addition to higher levels of DPP-4 in patients who respond to treatment, and higher levels of DPP-4 in UC patients (1467 (1301) -1641) vs 1211 (1011-1448) ng / mL;P -lt;0.001), while CD patients (1385 s 1185-1592) vs 1134 s 975-1469 sng / mL;P s 0.015).
Serum DEP-4 can be used as a non-invasive biomarker for IBD activity and biotherapy reactions and as a predictive indicator of therapeutic escalation, especially when used in combination with other biomarkers.
.