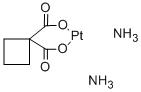
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Androgen receptor (AR) signaling inhibitors (ARSIs), including abiraterone acetate (AA), enzalutamide, apalutamide and darolutamide, can improve male metastatic castration-sensitive prostate cancer (CSPC) and Potential tolerate the survival of prostate cancer (CRPC)
Androgen receptor (AR) signaling inhibitors (ARSIs), including abiraterone acetate (AA), enzalutamide, apalutamide and darolutamide, can improve male metastatic castration-sensitive prostate cancer (CSPC) and Potential tolerate the survival of prostate cancer (CRPC)
Among them, 99 patients were used as the experimental set, and 2 prospective phase II clinical studies were used as the validation set (NCT01942837 [N=21] and NCT02025010 [N=27])
Among them, 99 patients were used as the experimental set, and 2 prospective phase II clinical studies were used as the validation set (NCT01942837 [N=21] and NCT02025010 [N=27])
Features of C1 and C2
The median OS of patients in the C2 and C1 groups was 8.
The median OS of patients in the C2 and C1 groups was 8.
OS
In multivariate analysis (MVA), after adjusting for patient age, CRPC and CSPC, NEPC and CSPC, PSA, visceral metastasis, and AR-status, C2 was a shorter independent prognostic factor for OS
In multivariate analysis (MVA), after adjusting for patient age, CRPC and CSPC, NEPC and CSPC, PSA, visceral metastasis, and AR-status, C2 was a shorter independent prognostic factor for OS
Prognostic factors
A total of 48 patients in the two clinical studies were used as validation sets, of which 10 patients were identified as Cluster2 (C2)
A total of 48 patients in the two clinical studies were used as validation sets, of which 10 patients were identified as Cluster2 (C2)
The validation set evaluates the prognosis of C1 and C2
In summary, studies have shown that the transcription profile detected in tumor circulating cells (CTCs) by liquid biopsy can be used as an independent prognostic marker other than AR-V7 in patients with metastatic prostate cancer, and can be used to identify multiple ARSI resistance mechanisms.
In summary, studies have shown that the transcription profile detected in tumor circulating cells (CTCs) by liquid biopsy can be used as an independent prognostic marker other than AR-V7 in patients with metastatic prostate cancer, and can be used to identify multiple ARSI resistance mechanisms.
Original source:
Sperger JM, Emamekhoo H, McKay RR, et al.
Prospective Evaluation of Clinical Outcomes Using a Multiplex Liquid Biopsy Targeting Diverse Resistance Mechanisms in Metastatic Prostate Cancer.
J Clin Oncol.
2021 Jul 1:JCO2100169.
doi: 10.
1200/JCO.
21.
00169.
Epub ahead of print.
PMID: 34197212.
Prospective Evaluation of Clinical Outcomes Using a Multiplex Liquid Biopsy Targeting Diverse Resistance Mechanisms in Metastatic Prostate Cancer.
J Clin Oncol.
2021 Jul 1:JCO2100169.
doi: 10.
1200/JCO.
21.
00169.
Epub ahead of print.
PMID: 34197212.
Leave a message here