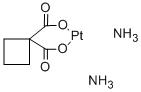
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
In a multi-center, multi-cohort, non-randomized, open-label phase II study (NCT01336634), the ORR of patients receiving dabrafenib monotherapy (cohort A) was 33% (95% CI, 23%) -45%), the PFS is 5.
In a multi-center, multi-cohort, non-randomized, open-label phase II study (NCT01336634), the ORR of patients receiving dabrafenib monotherapy (cohort A) was 33% (95% CI, 23%) -45%), the PFS is 5.
The primary endpoint is the overall response rate (ORR) assessed by the investigator based on the efficacy evaluation criteria for solid tumors version 1.
The primary endpoint is the overall response rate (ORR) assessed by the investigator based on the efficacy evaluation criteria for solid tumors version 1.
Cohort B eventually enrolled 57 patients, and cohort C enrolled 36 patients
At the time of data truncation, the median follow-up time of cohort B (57 cases) and cohort C (36 cases) were 16.
ORR of queue B
The DOR of patients in cohort B and cohort C were 9.
DoR
The median PFS (95% CI) of patients in cohort B and cohort C was 10.
The median PFS (95% CI) of patients in cohort B and cohort C was 10.
PFS
The most common AEs (any grade) that occurred in ≥30% of patients were fever (56%), nausea (51%), vomiting (41%), dry skin (39%), peripheral edema (38%), Diarrhea (37%), decreased appetite (33%) and cough (31%)
.
The most common grade ≥3 adverse reactions that occurred in ≥5% of patients were hypertension, hyponatremia, neutropenia, fever, dyspnea, anemia, and elevated alanine aminotransferase
.
In summary, the study shows that dabrafenib + trametinib has sustained clinical benefits in the treatment of metastatic BRAF V600E mutant non-small cell lung cancer, and its safety is controllable, regardless of the previous treatment
.
.
This study shows that dabrafenib + trametinib has sustained clinical benefit in the treatment of metastatic BRAF V600E mutant non-small cell lung cancer, and its safety is controllable, regardless of the previous treatment
.
This study shows that dabrafenib + trametinib has sustained clinical benefit in the treatment of metastatic BRAF V600E mutant non-small cell lung cancer, and its safety is controllable, regardless of the previous treatment
.
Original source:
Original source:Planchard D, Besse B, Groen HJM, et al.
Phase 2 study of dabrafenib plus trametinib in patients with BRAF V600E-mutant metastatic non-small cell lung cancer: Updated 5-year survival rates and genomic analysis.
J Thorac Oncol.
2021 Aug 26:S1556-0864(21)02403-5.
doi: 10.
1016/j.
jtho.
2021.
08.
011.
Epub ahead of print.
PMID: 34455067.
Phase 2 study of dabrafenib plus trametinib in patients with BRAF V600E-mutant metastatic non-small cell lung cancer: Updated 5-year survival rates and genomic analysis.
J Thorac Oncol.
2021 Aug 26:S1556-0864(21)02403-5.
doi: 10.
1016/j.
jtho.
2021.
08.
011.
Epub ahead of print.
PMID: 34455067.
Leave a message here