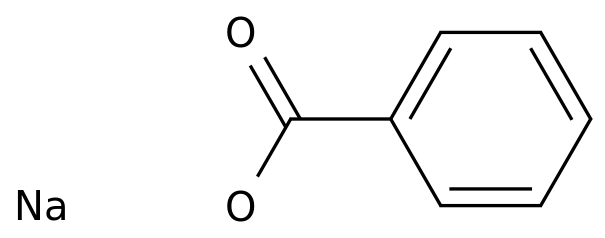
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
In this one-arm non-comparative trial, 13 patients with severe Staphylococcus acuity infection in an Australian hospital were given intravenously with a production practice of three strains of Mistococcus seedling preparation (AB-SA01) as an auxiliary treatment.
AB-SA01 was intravenously injected twice a day for 14 d, monitoring the subjects' clinical, hematological and blood biochemical parameters, and lasting 90 d, with the main result of assessing safety and tolerance (i.e. pain and redness at the infusion site and systemic adverse reactions such as fever, tinge, low blood pressure, diarrhea or abdominal pain, and the occurrence of kidney or liver dysfunction).
results, there were no reports of adverse reactions, and the results showed that AB-SA01, which was given in this manner, was safe in severe Staphylococcus aurus infections, including infectious endoarthritis and sepsis shock.
In the future, controlled trials will be required to determine the efficacy of AB-SA01, but there will be no phage resistance in the body, and measurements of bacterial and phage dynamics in blood samples suggest that 12-hour re-supply of 109 plaque-forming units may be a reasonable basis for further study.
.