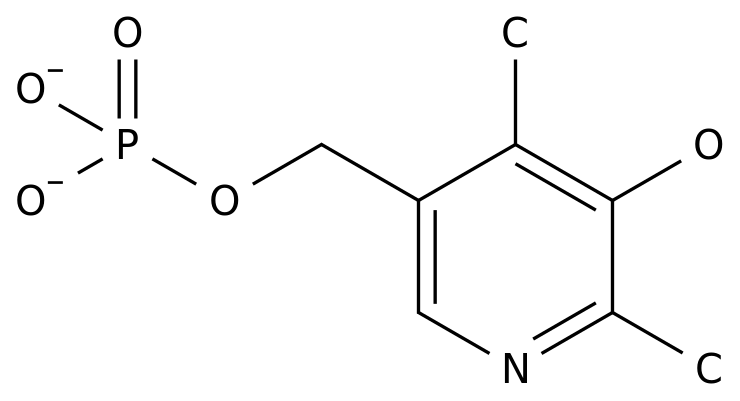
Paracetamol sodium for injection is accepted
-
Last Update: 2020-04-03
-
Source: Internet
-
Author: User
Search more information of high quality chemicals, good prices and reliable suppliers, visit
www.echemi.com
According to the website of the State Food and Drug Administration (CFDA) on April 2, the six kinds of chemical drugs used for injection, which are declared to be produced by orsacom, have been accepted At present, only the original research drugs are on sale in China The data showed that parecoxib sodium for injection was mainly used for the short-term treatment of postoperative pain, and it can be used for the treatment of moderate or severe postoperative acute pain, with quick effect, long-lasting effect and strong analgesic effect The original research drug is Pfizer's "special resistance", which entered the Chinese market in 2008 According to CFDA, apart from aosaikang, at present, only Qilu pharmaceutical, Hengrui pharmaceutical and Kelun pharmaceutical apply for the approval of the product in China Aosaikang is mainly engaged in the production and sales of digestive, anti-tumor and other drugs The key product is the digestive drug "Aoxikang" (omeprazole sodium for injection) Aoxikang accounts for more than 50% of the company's operating revenue and gross profit, and the company's leading products are too concentrated.
This article is an English version of an article which is originally in the Chinese language on echemi.com and is provided for information purposes only.
This website makes no representation or warranty of any kind, either expressed or implied, as to the accuracy, completeness ownership or reliability of
the article or any translations thereof. If you have any concerns or complaints relating to the article, please send an email, providing a detailed
description of the concern or complaint, to
service@echemi.com. A staff member will contact you within 5 working days. Once verified, infringing content
will be removed immediately.