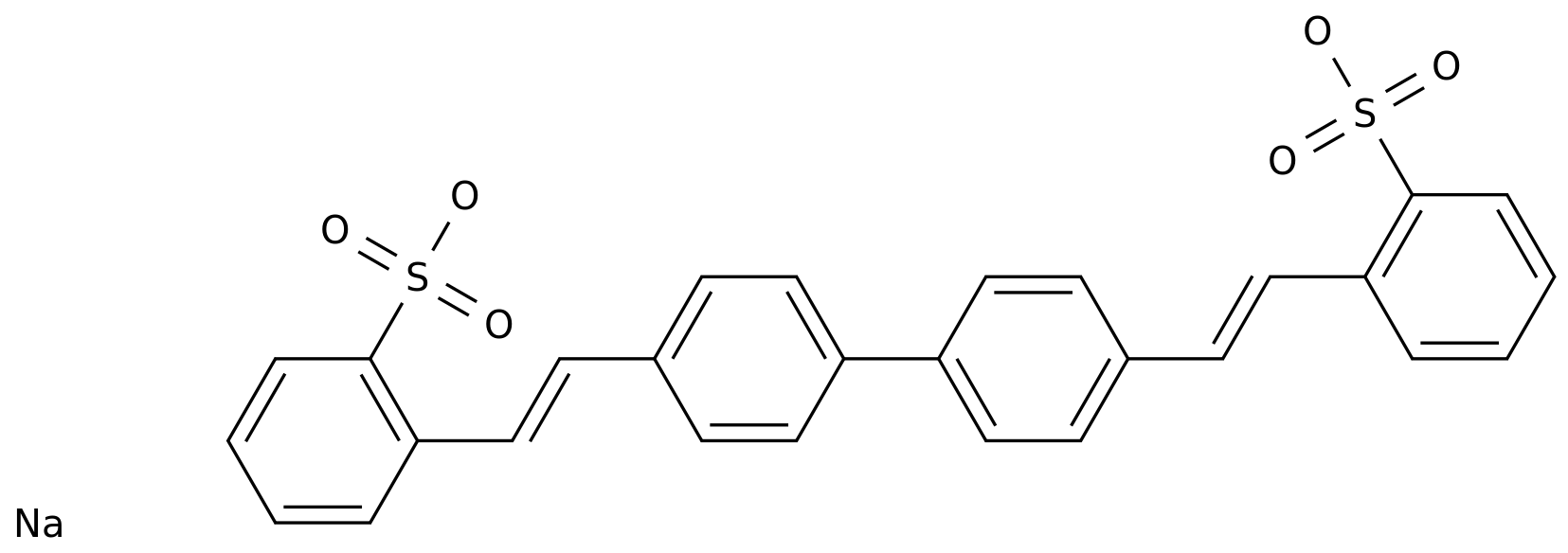
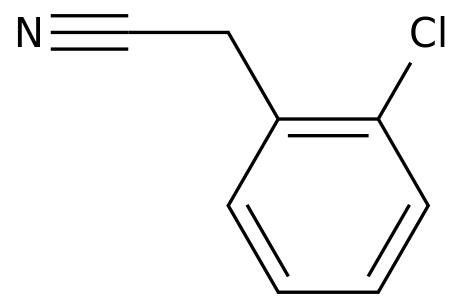
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
German and Finnish scientists wrote in the latest issue of "Science" that they have synthesized a unique new type of carbon atom network.
The new form of carbon is like graphene, only one atom thick, but its atomic structure and electronic properties are similar to those of graphite.
Alkenes are completely different and are expected to find their use in areas such as batteries.
? Carbon exists in various forms, such as diamond, graphite, and graphene.
Graphene is the thinnest material known so far, and its unique "character" makes it hopeful that it can be used in many applications such as electronics.
In graphene, each carbon atom is connected to three other carbon atoms to form a honeycomb hexagon.
Theoretical studies have shown that carbon atoms can also be arranged in other network patterns, but scientists have not yet discovered these predicted networks.
In the latest research, scientists from Marburg University and Aalto University discovered a new carbon atom network and named it the biphenylene network.
In this new form of carbon material, carbon atoms are connected to each other to form squares, hexagons and octagons, which are then combined into an ordered lattice.
The researchers also confirmed the unique structure of this network with a high-resolution scanning probe microscope and found that its electronic properties are completely different from graphene.
? They said that graphene is a semiconductor, but this new type of carbon has metallic properties.
Professor Michael Gottfried of the University of Marburg said: “The narrow stripes on the new carbon network behave like metals and can be used as wires for future carbon-based electronic devices.
” The lead author of the study, Qitang Fan of the University of Marburg (transliteration) ) Said: "This new type of carbon network can also be used as a positive electrode material for lithium-ion batteries.
Compared with the current graphene-based materials, the new material has a larger lithium storage capacity.
"? The researchers assembled carbon-containing molecules in smooth gold.
This new material is made on the surface: carbon-containing molecules first form chains of connected hexagons, which are then joined together to form squares and octagons.
Before the connection, only the same type of chains will gather on the gold surface to form an ordered combination, which is crucial for the formation of new carbon materials, because the reactions between different types of chains will only produce graphene.
Yan Linghao (transliteration) of Aalto University explained: "Our unique feature is the use of molecular precursors.
We adjust these molecular precursors to produce biphenylene instead of graphene.
"? At present, the research team is committed to research and development.
Larger thin slices of new materials to explore its application potential.
They also said that the new synthesis method is expected to give birth to other new types of carbon networks.
? The emergence of graphene opens the door to the world of two-dimensional materials and refreshes scientists' understanding of the "dimensionality" of material materials.
Since a single layer of carbon atoms can form graphene, in theory, other substances may also be single-layered atoms Forms are arranged and form a new two-dimensional material.
The latest research has brought new inspiration: changing the arrangement of single-layer carbon atoms can form another two-dimensional material.
Is the same with other materials? If this is the case, then the boundaries of the world of two-dimensional materials will be greatly expanded.
The new form of carbon is like graphene, only one atom thick, but its atomic structure and electronic properties are similar to those of graphite.
Alkenes are completely different and are expected to find their use in areas such as batteries.
? Carbon exists in various forms, such as diamond, graphite, and graphene.
Graphene is the thinnest material known so far, and its unique "character" makes it hopeful that it can be used in many applications such as electronics.
In graphene, each carbon atom is connected to three other carbon atoms to form a honeycomb hexagon.
Theoretical studies have shown that carbon atoms can also be arranged in other network patterns, but scientists have not yet discovered these predicted networks.
In the latest research, scientists from Marburg University and Aalto University discovered a new carbon atom network and named it the biphenylene network.
In this new form of carbon material, carbon atoms are connected to each other to form squares, hexagons and octagons, which are then combined into an ordered lattice.
The researchers also confirmed the unique structure of this network with a high-resolution scanning probe microscope and found that its electronic properties are completely different from graphene.
? They said that graphene is a semiconductor, but this new type of carbon has metallic properties.
Professor Michael Gottfried of the University of Marburg said: “The narrow stripes on the new carbon network behave like metals and can be used as wires for future carbon-based electronic devices.
” The lead author of the study, Qitang Fan of the University of Marburg (transliteration) ) Said: "This new type of carbon network can also be used as a positive electrode material for lithium-ion batteries.
Compared with the current graphene-based materials, the new material has a larger lithium storage capacity.
"? The researchers assembled carbon-containing molecules in smooth gold.
This new material is made on the surface: carbon-containing molecules first form chains of connected hexagons, which are then joined together to form squares and octagons.
Before the connection, only the same type of chains will gather on the gold surface to form an ordered combination, which is crucial for the formation of new carbon materials, because the reactions between different types of chains will only produce graphene.
Yan Linghao (transliteration) of Aalto University explained: "Our unique feature is the use of molecular precursors.
We adjust these molecular precursors to produce biphenylene instead of graphene.
"? At present, the research team is committed to research and development.
Larger thin slices of new materials to explore its application potential.
They also said that the new synthesis method is expected to give birth to other new types of carbon networks.
? The emergence of graphene opens the door to the world of two-dimensional materials and refreshes scientists' understanding of the "dimensionality" of material materials.
Since a single layer of carbon atoms can form graphene, in theory, other substances may also be single-layered atoms Forms are arranged and form a new two-dimensional material.
The latest research has brought new inspiration: changing the arrangement of single-layer carbon atoms can form another two-dimensional material.
Is the same with other materials? If this is the case, then the boundaries of the world of two-dimensional materials will be greatly expanded.