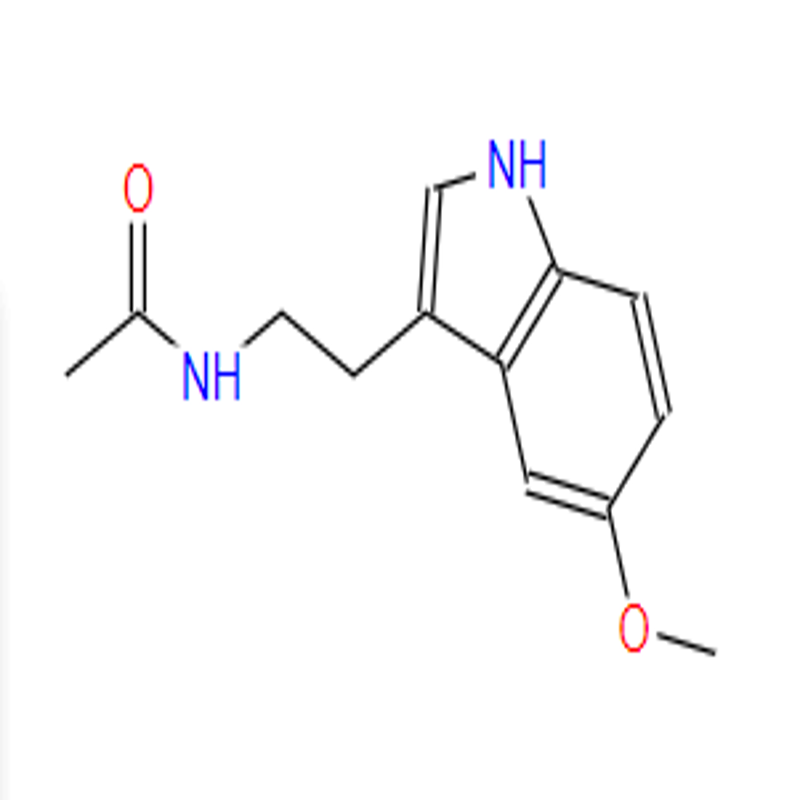
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
▎WuXi AppTec Content Team Editor On February 16, 2022, Equillium announced the acquisition of Bioniz Therapeutics
.
Bioniz's lead product candidates are multispecific inhibitors that simultaneously target multiple clinically validated disease-driving cytokines, including potential "first-in-class" therapies BNZ-1 and BNZ-2 in clinical development, aimed at Addressing unmet needs in a range of immuno-inflammatory indications
.
Through this acquisition, Equillium's immunology R&D pipeline and technology platform include: BNZ-1: a potential "first-in-class" trispecific inhibitor of leukocytes, inflammatory cytokines associated with a variety of diseases Interleukin (IL)-2, IL-9 and IL-15
.
BNZ-1 has completed its clinical proof-of-concept as a novel cytokine inhibitor through a Phase 1/2 clinical trial in patients with cutaneous T-cell lymphoma (CTCL), showing good tolerability and safety profile, and resulted in clinically meaningful improvements in mSWAT scores for patients
.
Equillium plans to first focus further development of BNZ-1 on alopecia areata for which there is no approved drug
.
BNZ-2: a potential "first-in-class" selective inhibitor of IL-15 and IL-21
.
Considerable translational work has been performed with BNZ-2 supporting its potential use in the treatment of a variety of gastrointestinal diseases, with IL-15 and IL-21 acting synergistically to drive celiac disease and other inflammatory bowel diseases, and liver disease science
.
The therapy is ready for Phase 1 clinical trials, with a proof-of-concept evaluation planned in celiac disease patients
.
BNZ-2 is currently formulated for subcutaneous administration and is designed to address the unmet needs of patients who continue to experience symptoms despite trying to maintain a gluten-free diet
.
Discovery Platform: Bioniz's flexible, structure-based technology platform combined with deep insights into cytokine pathways and computational modeling creates discovery capabilities that result in highly differentiated immunology products
.
Its discovery pipeline includes BNZ-3, a preclinical drug candidate that is a stable peptide designed for oral administration and local action in the gut to treat gastrointestinal inflammation
.
▲Bioniz product pipeline (Image source: Bioniz's official website) "This acquisition is transformative for Equillium
.
" Mr.
Bruce Steel, CEO of Equillium, said, "We look forward to advancing these molecules into later stages of development to provide Innovative therapies for patients with severe immune-inflammatory diseases
.
"Reference: [1] Equillium Acquires Bioniz Therapeutics Significantly Expanding Pipeline of Novel Immunomodulatory Drug Candidates.
Retrieved February 16, 2022, from https:// -Acquires-Bioniz-Therapeutics-Significantly-Expanding-Pipeline-of-Novel-Immunomodulatory-Drug-Candidates/default.
aspx Disclaimer: The WuXi AppTec content team focuses on introducing global biomedical health research progress
.
This article is for information exchange purposes only , the opinions in this article do not represent WuXi AppTec's position, nor do they support or oppose the opinions expressed in the article
.
This article is not a treatment plan recommendation
.
If you need treatment plan guidance, please go to a regular hospital for treatment
.
.
Bioniz's lead product candidates are multispecific inhibitors that simultaneously target multiple clinically validated disease-driving cytokines, including potential "first-in-class" therapies BNZ-1 and BNZ-2 in clinical development, aimed at Addressing unmet needs in a range of immuno-inflammatory indications
.
Through this acquisition, Equillium's immunology R&D pipeline and technology platform include: BNZ-1: a potential "first-in-class" trispecific inhibitor of leukocytes, inflammatory cytokines associated with a variety of diseases Interleukin (IL)-2, IL-9 and IL-15
.
BNZ-1 has completed its clinical proof-of-concept as a novel cytokine inhibitor through a Phase 1/2 clinical trial in patients with cutaneous T-cell lymphoma (CTCL), showing good tolerability and safety profile, and resulted in clinically meaningful improvements in mSWAT scores for patients
.
Equillium plans to first focus further development of BNZ-1 on alopecia areata for which there is no approved drug
.
BNZ-2: a potential "first-in-class" selective inhibitor of IL-15 and IL-21
.
Considerable translational work has been performed with BNZ-2 supporting its potential use in the treatment of a variety of gastrointestinal diseases, with IL-15 and IL-21 acting synergistically to drive celiac disease and other inflammatory bowel diseases, and liver disease science
.
The therapy is ready for Phase 1 clinical trials, with a proof-of-concept evaluation planned in celiac disease patients
.
BNZ-2 is currently formulated for subcutaneous administration and is designed to address the unmet needs of patients who continue to experience symptoms despite trying to maintain a gluten-free diet
.
Discovery Platform: Bioniz's flexible, structure-based technology platform combined with deep insights into cytokine pathways and computational modeling creates discovery capabilities that result in highly differentiated immunology products
.
Its discovery pipeline includes BNZ-3, a preclinical drug candidate that is a stable peptide designed for oral administration and local action in the gut to treat gastrointestinal inflammation
.
▲Bioniz product pipeline (Image source: Bioniz's official website) "This acquisition is transformative for Equillium
.
" Mr.
Bruce Steel, CEO of Equillium, said, "We look forward to advancing these molecules into later stages of development to provide Innovative therapies for patients with severe immune-inflammatory diseases
.
"Reference: [1] Equillium Acquires Bioniz Therapeutics Significantly Expanding Pipeline of Novel Immunomodulatory Drug Candidates.
Retrieved February 16, 2022, from https:// -Acquires-Bioniz-Therapeutics-Significantly-Expanding-Pipeline-of-Novel-Immunomodulatory-Drug-Candidates/default.
aspx Disclaimer: The WuXi AppTec content team focuses on introducing global biomedical health research progress
.
This article is for information exchange purposes only , the opinions in this article do not represent WuXi AppTec's position, nor do they support or oppose the opinions expressed in the article
.
This article is not a treatment plan recommendation
.
If you need treatment plan guidance, please go to a regular hospital for treatment
.