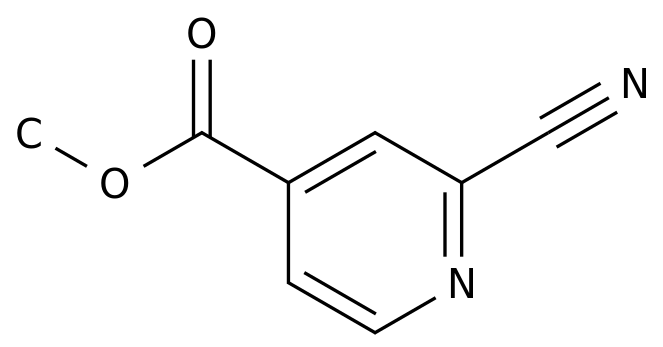
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
In order to strengthen the supervision and management of the quality of medical devices, to ensure that medical device production and operation enterprises and user units in the transport and storage process to make products meet the specific temperature requirements of their instructions and labels, in accordance with the Regulations on the Supervision and Administration of Medical Devices (State Council Order No. 650), the Medical Devices Management Measures (State Food and Drug Administration Order No. 8) and the Medical Device Use Quality Supervision and Administration (State Food and Drug Administration Order No. 18), the State Food and Drug Administration has formulated the "Cold Chain"
this announcement.Article
The Management Guide for Cold Chains of Medical Devices (Transport, Storage
This Guide is formulated in accordance with the requirements of relevant laws and regulations, such as the Regulations on the Supervision and Administration of Medical Devices, in order to ensure that medical device production and operation enterprises and user units in the process of transportation and storage make the products meet the specific temperature requirements indicated in their instructions and labels.
second cold chain management medical device refers to the medical device that needs to be refrigerated and frozen in accordance with the instructions and labeling requirements during transportation and storage. This guide applies to the quality management of medical device transportation and storage by medical device production and operation enterprises and user units.article
Personnel engaged in the receipt, acceptance, storage, inspection, storage, storage and transportation of cold chain management medical devices shall be trained in refrigeration, refrigeration-related laws and regulations, professional knowledge, work system and standard operating procedures before they are qualified for employment.
article 4 Medical device manufacturers and wholesale enterprises shall be equipped with appropriate cold storage (cold storage or freezers) and refrigerated trucks or freezers (insulation boxes) and other facilities and equipment according to the variety and scale of production and operation.
medical device retail enterprises and user units shall be equipped with appropriate cold storage or refrigeration equipment (freezers or freezers, etc.) according to the type and size of operation and use.
5 of this article, the cold storage for the storage of medical equipment shall have the function of automatic temperature control, and the refrigeration capacity of the unit shall be suitable for the capacity of the cold storage. In order to ensure the continuous power supply of the refrigeration system, the cold storage should be equipped with a backup generator set or dual-loop power supply system.
storage shall be divided into areas to be examined, storage areas, return areas, packaging materials pre-cooling areas (places), etc., and marked clearly.
Article 6 Refrigerated vehicles used for the transportation of medical devices shall have automatic temperature control functions, the carriers shall be waterproof and closed, and there shall be space in the carriers to ensure adequate circulation of airflow.
Article 7 Refrigeration boxes (cabinets) shall be able to automatically adjust the temperature in the body of the box, and the insulation boxes shall be equipped with cold storage (heat) agents and insulation devices, and shall comply with the storage and transportation requirements indicated in the product instructions and labels.
8 of this article, cold storage and refrigerated vehicles used for the storage and transportation of medical devices shall be equipped with automatic temperature monitoring system (hereinafter referred to as temperature measurement system) to monitor temperature. The temperature measurement system should have the following functions:
(1) the measurement range, accuracy, resolution and other technical parameters of the temperature measurement system can meet the management needs, with continuous monitoring, continuous recording, data storage, display and alarm functions.
(ii) The operation process of cold storage and refrigerated truck equipment is updated at least every 1 minute to measure the temperature data, the storage process automatically records real-time temperature data at least every 30 minutes, and the transportation process automatically records real-time temperature data at least every 5 minutes.
(iii) When the monitoring temperature reaches the set threshold or exceeds the specified range, the temperature measurement system can achieve sound and light alarm, and at the same time realize the communication mode such as SMS to send an alarm message to at least 2 designated personnel immediately.
each (station) independent cold storage, refrigeration vehicle should be based on the verification conclusions set, install at least 2 temperature measuring point terminals. Temperature measuring terminals and temperature measuring equipment should be calibrated or checked at least once a year.
, insulation or other refrigeration equipment should be equipped with automatic temperature recording and storage equipment.
. Article 9 Refrigeration, refrigerated trucks, refrigerated containers, insulation boxes and temperature measurement systems shall be verified for pre-use verification, periodic verification and deactivated for longer than the prescribed time limit. Unverified facilities and equipment shall not be used in the transportation and storage process of cold chain management medical devices.
(i) to establish and form a validation management document, including verification programs, standards, reports, evaluation, deviation processing and preventive measures.
(2) determine a reasonable continuous verification time according to the verification object to ensure the full, effective and continuous verification data.
(3) The temperature measurement equipment used for verification shall be calibrated or verified by a qualified measurement institution, and the calibration or verification certificate (copy) shall be used as a necessary attachment to the verification report, and the verification data shall be true, complete, valid and traceable.
(4) The relevant facilities and equipment are used correctly and reasonably according to the parameters and conditions determined by the verification.
Article 10 When carrying out cold chain management medical device receipt, the mode of transportation, in-transit temperature, time of shipment and time of delivery shall be verified and recorded, and the temperature records of the period of sale shall also be verified for the products returned after sale. Those who meet the requirements shall be moved into the waiting area in the cold storage in time; those who do not meet the temperature requirements shall be refused and recorded accordingly.
11th, the use of cold storage cold chain management medical equipment, should be in the cold storage for acceptance.
inspection and acceptance personnel shall check the status of the products and make a good record in accordance with the requirements of Articles 38, 39 or the Measures for the Supervision and Administration of the Use of Medical Devices.
Article 12 Cold Chain Management Medical Devices shall be stored and inspected in accordance with the requirements of the product description or label marking during the storage period, and the packaging, labeling, appearance and temperature status of the stored cold chain management medical devices shall be examined and recorded.
the air-out port of the refrigeration unit in the cold storage shall be avoided, and a reasonable storage area shall be determined according to the cold storage verification report.
the discharge of medical devices under the control of article 13 of the 13th cold chain, a special person shall be responsible for the review, packing and sealing, and loading and release of the vehicle code.
Where a medical device is managed by using a cold storage box or insulation box for transportation of cold chains, the standard operating procedures for packaging shall be formulated in accordance with the parameters and conditions determined by the verification, and the packing and sealing operation shall meet the following requirements:
(1) the refrigerated box, insulation box pre-cooling or preheating shall be carried out before packing.
(ii) reasonablely equipped with refrigerants appropriate to temperature control and transport time limits in the insulation box.
(iii) The refrigeration box starts the refrigeration function and the temperature measurement equipment (the insulation box starts the temperature measurement equipment), inspects the equipment to operate normally, and after reaching the specified temperature, packs the product.
(iv) According to the verification conclusions of temperature control of refrigerants and products, if necessary, the product should be isolated from refrigerants such as refrigerants using a heat insulation device.
(5) The packaging, packing and sealing of cold chain management medical devices shall be completed in accordance with the temperature range indicated in the product instructions and labels.
Article 14 Where a medical device is managed by a transport cold chain, reasonable means of transport and temperature control shall be selected according to the quantity, distance, time and temperature requirements of the products transported, and the temperature of the external environment, so as to ensure that the temperature control during transport meets the requirements.
Article 15 Where a refrigerated truck is used to transport cold chain management medical devices, the following requirements shall be met:
(1) the refrigeration function and temperature measurement equipment shall be activated in advance, and the cabin shall be pre-cooled to the specified temperature.
(2) According to the verification report, determine the code release mode and area of the product in the refrigerated compartment, the code release height shall not exceed the lower edge of the air discharge of the refrigeration unit, and ensure the normal circulation of the air flow and the uniform distribution of the temperature.
(iii) cold chain management medical equipment installed, timely closure of the car door, check the closure of the door.
(4) check the health of temperature control equipment and temperature measurement equipment, normal operation can be started.
(v) Cold chain management medical devices in the loading and unloading process, measures should be taken to ensure that the temperature meets the requirements of the product description and labeling.
the 16th cold chain to manage the delivery of medical devices, the temperature of refrigerated vehicles, refrigerated containers and insulation boxes shall be checked and recorded. Upon delivery, the receiving unit shall be provided with a temperature record of the entire period of shipment. article
Where other units are entrusted with the transportation of cold chain management medical devices, the qualifications and capabilities of the carrier shall be audited and the commissioned transportation agreement shall be signed, at least in accordance with the following requirements:
(1) request the carrier's transport qualification documents, transport facilities equipment and transport management monitoring system verification documents, carrier qualification certificates, transport process temperature control and monitoring system verification documents and other relevant information.
(2) Conduct pre-commissioned and regular audits of the carrier's transportation facilities equipment, personnel qualifications, quality assurance capabilities, safe transportation capabilities, risk control capabilities, etc., and audit reports for archival review.
(iii) The contents of the commissioned transport agreement shall include: the operating procedures of the transport standards established by the carrier, the requirements for temperature control and real-time monitoring during transport, the requirements for time limits in transit and the responsibility for quality and safety during transport.
(4) If necessary, according to the qualifications and conditions of the carrier, the entrusting party may review and assess the relevant personnel of the carrier and the equipment of the transport facilities.
entrusting other units to store cold chain management of medical devices, the entrusted enterprise shall comply with the requirements of Article 31 of the Code of Quality Management of Medical Device Operations.
19 Production and operation enterprises and user units shall formulate emergency plans for temperature control of cold chain management medical devices during storage and transportation, and verify emergency plans. In case of power failure, abnormal climate, equipment failure, traffic accident and other accidents or emergencies in the course of storage and transportation, effective measures can be taken in time to prevent the temperature from getting out of control due to abnormal emergencies.
article 20 this guide shall be interpreted by the State Administration of Food and Drug Administration.
Article 21 of this Guide shall be implemented from the date of publication. (State Administration of Food and Drug Administration)