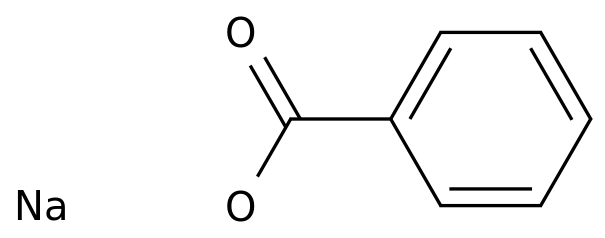
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
The Serum Institute of India (SII) said it would obtain authorization from AstraZeneca and Novavax to produce the COVID-19 vaccine.
SII, the
, said that up to 100 million doses of vaccines will be produced in India and low- and middle-income countries as early as the first half of the year, using funds from Gavi, the Vaccine Alliance and the Bill and Melinda Gates Foundation. "To ensure maximum immunization coverage and curb pandemics, it is important to ensure that the world's most remote and poorest countries have access to affordable treatment and preventive measures," said Adar Poonawalla, chief executive of
SII.
Gavi's board recently approved a list of 92 countries, the countries on the list will be supported by the organization's COVAX Advanced Market Commitment (AMC), which aims to ensure rapid and fair access to the COVID-19 vaccine worldwide.
specific, COVAX aims to deliver 2 billion doses of the approved COVID-19 vaccine by the end of 2021.
early data from the Phase I/II clinical trial, published last month in The Lancet, showed that AstraZeneca's vaccine AZD1222, developed in collaboration with Oxford University, raised neutralizing antibody levels and T-cell levels against the SARS-CoV-2 virus. similar
ly, Novavax recently reported preliminary results from a Phase I/II study showing that its recombinant coronavirus vaccine candidate NVX-CoV2373 also triggered neutralizing antibodies in healthy adults, as well as cell immune responses from some of the participants.
.