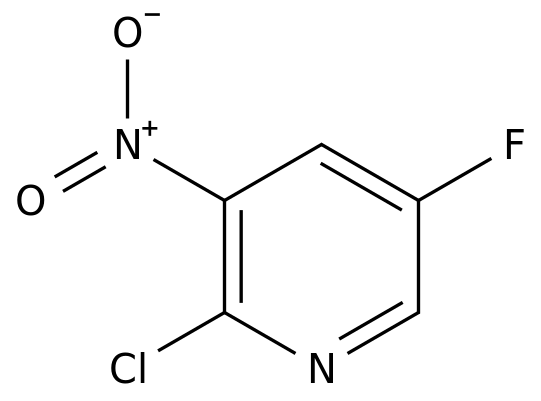
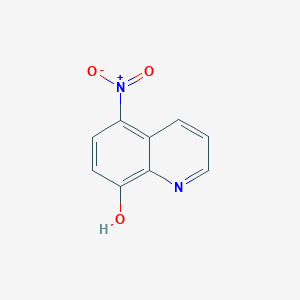
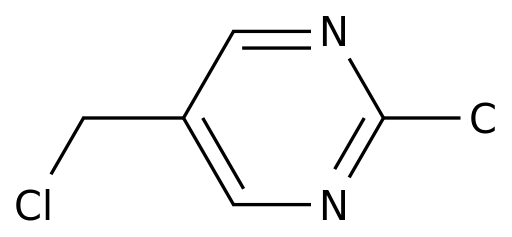
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
On December 29, the official website of the State Drug Administration issued an announcement on the revision of the instruction manual for the injection of mentatinine birdine, the instruction manual for the injection (including the injection of methionine, the injection of methionine, the prescription of holsteine osine), "precautions", "taboos", "pregnant and lactating women's medication", "children's medicine", "old age medication" to revise.
The specific announcement is as follows: The State Drug Administration's announcement on the revision of the instruction manual for the injection of mentamate birdine (No. 121 of 2020) in order to further ensure the safety of public drug use, the State Drug Administration decided to the mendionine birdine injection (Including mentatinine birdine injection, injection of methionine birdine) instruction manual "adverse reactions", "precautions", "taboos", "pregnant and lactating women medication", "children's medicine", "old age medication" items are revised.
The relevant matters will now be announced as follows: First, all production enterprises of mentamate birdine injections shall, in accordance with the Measures for the Administration of Drug Registration and other relevant provisions, in accordance with the revised requirements of the instructions for the injection of mendionine birdine (see annex), put forward a supplementary application for the revision of the instructions, before January 28, 2021 reported to the provincial drug regulatory authorities for the record.
of the amendment relates to the drug label, it shall be revised together, and the instructions and other contents of the label shall be consistent with the original approval content.
to replace all factory-issued drug instructions and labels within 9 months of filing the supplementary application.
The above-mentioned production enterprises for the occurrence of new adverse reactions should conduct in-depth research on the mechanism of occurrence of new adverse reactions, take effective measures to do a good job in the use and safety of publicity and training, related to drug safety content changes should immediately and appropriately notify the drug management and use units, guide physicians, pharmacists reasonable drug use.
. Clinicians and pharmacists shall carefully read the revised contents of the instruction manual for the injection of methionine and, when selecting the drug, conduct a full benefit/risk analysis in accordance with the newly revised instruction manual.
, patients should strictly follow the doctor's instructions to take drugs, before taking drugs should carefully read the instructions.
this announcement.
: The revision of the instruction manual for the injection of methionine is required by the State Drug Administration on October 28, 2020