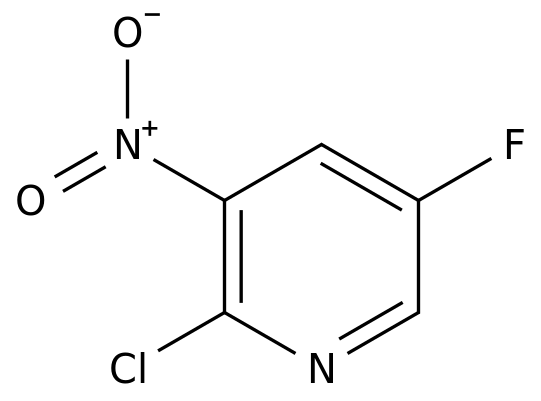
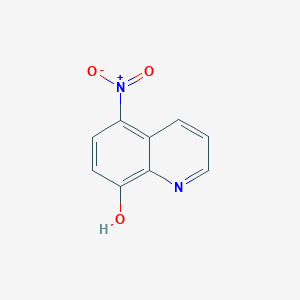
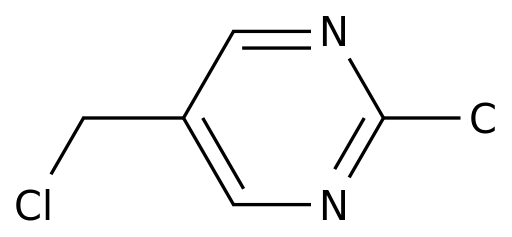
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
[
pharmacy network market analysis] It is reported that May 15, the National Insurance Bureau with the amount of the national procurement conference held in Shanghai Pharmaceutical Affairs, to discuss the State Central Purchasing-related content, including a second group of countries with start with the third installment of the ground situation and the amount of the purchase amount of procurementAt present, with the second batch of purchase with the amount gradually around the floor, with the third installment of the amount of procurement-related information has also been highly concerned about human medicineThe author briefly summarized according to industry sources comb, only for the industry referencewith the third installment of the amount of purchase and when to start?industry sources show, the third batch of statistics Jicai Drug List deadline to the end of June 2020, if progress smoothly, is expected to start in July, but still faces variablesMay 15 meeting, the experts say, whether the third installment of the Central Purchasing time later postponed it needs to be determinedIn the third series of national Jicai before landing, you may want to do first national Jicai three kinds of consumables, a few days before the National Insurance Bureau Yiba statistical tables sent to the countryWhetherround Jicai rules will change?industry sources, the third installment of the State Central Purchasing will follow the program in the second batch of good content, the selection of products ranging from fully competitive species, namely the original drug and had at least consistent evaluation of varieties " 1 + 2 "patternIndividual details will do fine-tuning, but is currently fine-tuning the specific content has not been fully determinedProbabilitywhich species into the Central Purchasing large?Earlier, the Thai Securities analysts believe that the 4 + 7 according to the pilot, to expand around 4 + 7, when the second group of countries of Central Purchasing, Central Purchasing varieties included in the basic assessment is too generic drug companies more variety, with the third installment of the amount of procurement is likely to continue to follow the three and older mode of competition, that is the original home +1 Review and two over and over, the number of entrepreneurs in the assessment are expected over two or more (excluding the original research) the varieties are likely included in the next batch of countries of Central PurchasingPing An Securities had expected the third round of the National Central Purchasing will start fast, too ≥3 family enterprise comment varieties included in probability Haitong Securities in a research report "compete in China - local multinational pharmaceutical companies and pharmaceutical companies contest" the analysis pointed out that the probability of a large amount of procurement was brought primarily sample hospitals Top100 best-selling drug, and had commentary variety of manufacturers, as indicated in accordance with Insurance Bureau, more popular varieties with possible inclusion volume purchases; furthermore, may be incorporated with a variety of non-pharmaceutical Top100 selling purchase amount, and had a variety of manufacturers commentary There pharmaceutical analysts believe that the new round with the amount of purchases could push the boundaries of conformity assessment, health insurance directory, medicines and supplies "The use of large, high prices, there is not falling space Jicai drugs may be incorporated into the Central Purchasing Non-Medicare species Jicai also possible, but will not become the focus of the Central Purchasing."
Haitong Securities analysts pointed out that the company can see the number of successful varieties, with the amount of procurement resources to focus on domestic head
Historical data proves that multinational pharmaceutical companies in the future to deal with the amount of purchases will become more active, to bring mature new round with the amount involved in drug procurement or multinational pharmaceutical companies will be the focus of the work at this stage heavy file recently released a new signal, a related paper on the main points of 2020 Pharmaceutical Affairs Division of the National Committee of Pharmaceutical Affairs Division Wei Jian issued outflow, signal causes the release of corporate concern files emphasis, to do national organizations with centralized purchasing selected clinical use of drugs work While comprehensively promote drug use monitoring The industry believes that the current special period, with a variety may be affected by the amount of procurement or distribution of drugs reserves, temporary out of stock For example, shortly before the Shanghai Sunshine Pharmaceutical procurement network "issued on the implementation of the second batch of national centralized drug purchasing organization selected results of work suggests (2)" that the second batch of 32 national organizations centralized drug procurement varieties selected, the Ogilvy sand Tan acetate tablets, tablets ambrisentan, polyethylene glycol 4000 powder, levocetirizine tablets four cultivars supply, delivery delays and other issues may be the beginning of the implementation, which olmesartan medoxomil is expected in June to gradually recovery of production capacity, the normal supply visible, Central Purchasing drugs in clinical work with some of the problems inevitably occur in use, and the drug is representing the Department of Justice issued its attention to this issue, and it shall be classified as 2020 priorities Central Purchasing companies need to participate in the supply side, good quality guarantee In addition, with the advancement of monitoring drug use, drug use in the clinic will no doubt be more stringent.