-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
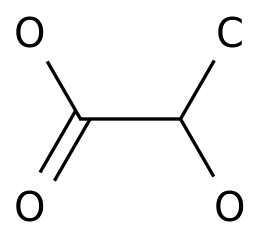
Ethyl acetate (ethyl acetate) is also known as ethyl acetate, the molecular formula is C 4 H 8 O 2 , the molecular weight is 88.
11, and the structural formula is
1.
Product performance
Colorless and transparent liquid with aromatic odor; melting point -83.
6°C, boiling point 77.
06°C, relative density (d 4 20 ) 0.
902, refractive index (n D 20 ) 1.
3719, flash point -3°C, auto-ignition temperature 426°C; in air Explosive limit 2.
2%-9%; soluble in chloroform , ethanol , ether , slightly soluble in water; forming an azeotrope with a boiling point of 70.
4℃ with water, and an azeotrope with a boiling point of 71.
8℃ with ethanol; vapor pressure 73×133.
3 Pa(20℃)
.
2.
Production principle
Industrially, sulfuric acid is used as a catalyst to make acetic acid and ethanol undergo an esterification reaction
.
3.
Process flow
4.
Technical formula (kg/t)
5.
Main equipment
Esterification kettle, washing pot, filter rectification kettle (rectification tower), liquid separator, storage tank
.
6.
Production process
Add 400kg of acetic acid, 375kg of ethanol, and 3kg of concentrated sulfuric acid to the enamel reaction kettle, stir, heat, and reflux for 6 hours.
Steam the crude ethyl acetate in the kettle and put it in the washing pot, and wash with the prepared 5% sodium chloride solution , And then adjust the pH of the material in the pot to 8 with a mixed aqueous solution of sodium hydroxide and sodium chloride
.
Then it was washed once with calcium chloride solution to separate the water layer
7.
Laboratory preparation method
Place a 250 mL round bottom flask containing 25 mL of ethanol under ice water cooling, and slowly add 25 mL of concentrated sulfuric acid
.
The mouth of the flask is equipped with a separatory funnel through a stopper, and the lower end is immersed in the liquid, and at the same time, a bend glass tube connected with a straight condenser is inserted into the stopper
8.
Product Standard
9.
Quality inspection
Assay: Accurately weigh about 1.
5g of the sample, add it to a conical flask, add 20mL of 0.
In GB3728, gas chromatography is used for content determination.
For specific operations, please refer to the national standard GB3728
.
10.
Product use
It is an important spice additive, which can be used as a component of flavoring, and can also be used as a flavor extractant for special modified alcohols
.
It is an excellent industrial solvent.
It can be used for nitrocellulose, ethyl fiber, chlorinated rubber and vinyl resin, cellulose acetate, cellulose acetate and synthetic rubber.
It can also be used for liquid nitrocellulose ink for copiers.
.
It can be used as a solvent for adhesives and a thinner for spray paint
.
It is the raw material for the manufacture of dyes and medicines
.
11.
Safety measures
(1) It is irritating to eyes, skin and mucous membranes
.
Toxicity: oral administration of the LD 50 of 5.
6 g / kg
.
The maximum allowable concentration of ethyl acetate in the workplace is 0.
04%
.
The equipment should be closed and the workshop should be well ventilated
.
Workers should wear protective equipment
.
(2) Packed in iron drums, stored in a dry and cool place, and stored and transported in accordance with the regulations for flammable and explosive dangerous goods
.
(3) This product is a first-class flammable product, and can form an explosive mixture with air, with an explosion limit of 2.
2% to 11.
5%
.
Related links: Production process and product use of 4-bromonaphthalene-1,8-dicarboxylic acid anhydride