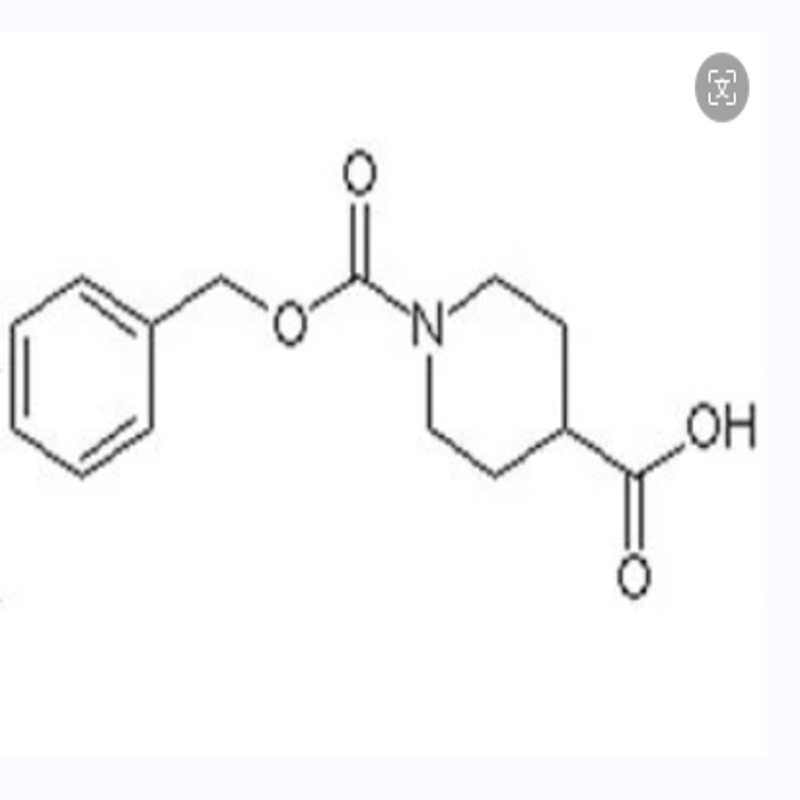
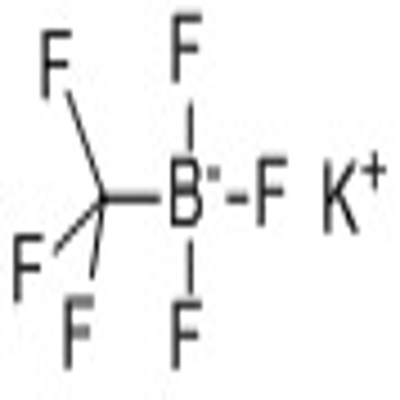
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
On July 17,, the FoodDrugAdministration (FDA) passed a safety label correction: warning that Evista; Eli Lilly and Coincrease the risk of stroke-related deathPreviously, the agency warned that women who use reloxifen increase the risk of venous thromboembolism (deep venous vascular embolism and pulmonary embolism) and now use the drug to increase the risk of stroke-related mortalityRandomized, double-blind and multi-country clinical trials were conducted in 10,101 postmenopausal women with coronary heart disease or who may be at risk of coronary heart diseaseCompared to the placebo group, the trial group, which used reloxifen for an average of 5.6 years, increased the mortality rate due to stroke (deaths: 59 (1.2%) vs39 (0.8%);However, there was no statistical difference in the incidence of stroke between the groups (249 (4.9%) vs224 (4.4%); Per 1,000 women per year: 9.5 vs8.6; harm ratio: 1.10; 95% confidence interval: 0.92 - 1.32; P .30), women's risk-benefit ratio of stroke, such as stroke history or transient ischemic episodes, atrial fibrillation,hypertensionor smoking, etcshould be considered aboveCardiovascular benefits were not observed in the study, so reloxifen should not be the preferred or secondary drug to prevent cardiovascular disease
Reloxifen is a drug used to prevent and treat osteoporosis in postmenopausal women