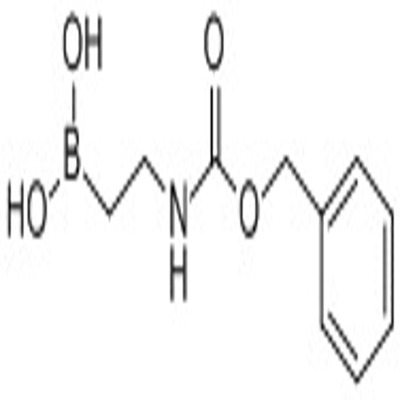
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Recently, the Office of the Guangxi Zhuang Autonomous Region Health and Health Commission announced the "July 2020 list of clinical applications of restricted medical technology" (with a complete directory at the end of the text).
, the Hai city health care committee has also issued the "Shanghai, medical technology clinical application management measures, the implementation of the Rules."
30 technologies, such as cardiovascular disease interventional diagnosis and treatment technology, artificial hip and knee replacement technology, neurovascular interventional diagnosis and treatment technology, were included in the "Shanghai Limited Clinical Application Medical Technology Directory".
In a number of restricted catalogues, many times involved in cardiovascular disease interventional diagnosis and treatment technology, which is related to stents, catheters, wire, embolism, puncture needles and other high-value supplies, replacement technology mainly involves artificial hip joints, artificial knee joints and artificial discs and other high-value consumables, 13 endoscopic diagnosis and treatment technology involves endoscopes, punctures and other types of medical devices.
medical technology is limited, resulting in a decrease in the number of operations, but also will inevitably lead to a decline in the use of hospital supplies and procurement.
is the restriction? The main reason for the limitation is security and effectiveness.
Taking the Shanghai Regulations as an example, medical technology in one of the following cases belongs to the "restriction technology": 1, the technical difficulty is high, the service ability of medical institutions, the level of personnel has higher professional requirements, the need to set restrictions;
the Shanghai Regulations, Shanghai has established a dynamic management mechanism to dynamically evaluate and adjust the catalogue of restricted technologies according to the actual situation.
relevant technologies are included in the catalogue, they will be strictly managed, and the Shanghai Regulations apply to all kinds of medical institutions and medical personnel at all levels in Shanghai to carry out clinical applications of medical technology.
, the clinical application management norms of restricted technologies will be formulated and issued by the Municipal Health and Health Commission or entrusted to professional organizations for publication, and reported to the National Health and Health Commission for the record.
For medical technologies that are not included in the list of prohibited and restricted technologies, medical institutions may, at their discretion, carry out clinical applications according to their own functions, tasks, technical capabilities, etc., and shall strictly manage the clinical applications of medical technologies carried out.
does not mean prohibiting some medical technologies from being included in the restricted list as restricted technologies, but restrictions do not mean prohibiting them.
In fact, in August 2018, the National Health and Health Commission issued the Measures for the Management of Clinical Application of Medical Technology, which requires medical institutions to conduct self-assessment in accordance with the relevant clinical application management norms for medical technology, and qualified to carry out clinical applications of restricted technologies.
a specific filing list:




![[4-methylsulfonyl-2-(trifluoromethyl)phenyl]boronic acid](https://file.echemi.com/fileManage/upload/goodpicture/20210823/m20210823171652704.jpg)