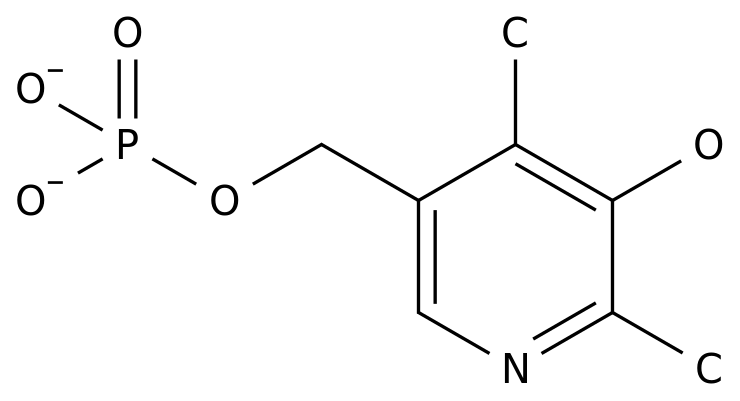
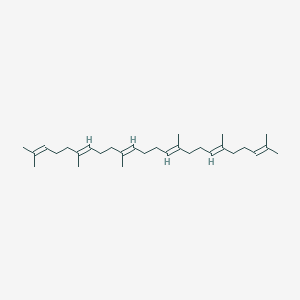
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Medical Network News on June 7th In China, the incidence and mortality of lung cancer are ranked first.
The reporter learned from Xinda Biotechnology on the 6th that the domestic innovative drug PD-1 inhibitor Daboshu has been approved by the National Medical Products Administration to combine chemotherapy in the first-line treatment of advanced squamous non-small cell lung cancer.
The reporter learned from Xinda Biotechnology on the 6th that the domestic innovative drug PD-1 inhibitor Daboshu has been approved by the National Medical Products Administration to combine chemotherapy in the first-line treatment of advanced squamous non-small cell lung cancer.
Medical experts pointed out that lung cancer is divided into small cell lung cancer and non-small cell lung cancer according to histological type.
Among them, cases of non-small cell lung cancer account for about 80% to 85% of lung cancer patients.
About 30% of Chinese patients with non-small cell lung cancer have squamous non-small cell lung cancer.
Among them, cases of non-small cell lung cancer account for about 80% to 85% of lung cancer patients.
About 30% of Chinese patients with non-small cell lung cancer have squamous non-small cell lung cancer.
Shanghai Pulmonary Medicine Hospital Professor Zhou Caicun oncology director, said the last two decades, progress of drug research and development of non-small cell lung cancer are mainly concentrated in non-squamous non-small cell lung cancer, squamous non-small cell lung cancer because of its unique epidemiology, organization Pathological and molecular biological characteristics, drug development is relatively slow, and patients lack effective drug treatment.
"Tumor, as the number one disease threatening human life and health , has always been a tough topic for researchers and clinicians.
Anti- tumor biologics are also an important research area in our product pipeline.
" said Dr.
Liu Yongjun, President of Cinda Bio, Dabo Shu has a complete layout in the field of squamous non-small cell lung cancer, providing new medication options for lung squamous cell carcinoma patients who previously lacked effective therapeutic drugs.
Anti- tumor biologics are also an important research area in our product pipeline.
" said Dr.
Liu Yongjun, President of Cinda Bio, Dabo Shu has a complete layout in the field of squamous non-small cell lung cancer, providing new medication options for lung squamous cell carcinoma patients who previously lacked effective therapeutic drugs.
It is reported that in Daboshu's layout for squamous non-small cell lung cancer, in addition to the first-line treatment approved this time, the key phase III clinical study of the second-line treatment also achieved positive results.
The listing application was also accepted by the State Drug Administration in January this year .
The listing application was also accepted by the State Drug Administration in January this year .
According to the R&D team, it is currently working hard to expand the scope of indications for Daboshu and has carried out more than 20 clinical studies on Daboshu worldwide.
Clinical research in the fields of colorectal cancer, gastric cancer, cholangiocarcinoma, cervical cancer, lymphoma, and melanoma is also underway.
Clinical research in the fields of colorectal cancer, gastric cancer, cholangiocarcinoma, cervical cancer, lymphoma, and melanoma is also underway.
It is worth mentioning that Daboshu is the first PD-1 in the country to enter the national medical insurance catalog.
In January this year, Cinda Bio responded to the Beijing Kangmeng Foundation's call to provide medical aid to cancer patients who were impoverished due to illness, and participated in the "Shuxinkeyi-Tumor Immunotherapy Patient Rescue Project" to effectively reduce the financial burden of patients.
(Finish)
In January this year, Cinda Bio responded to the Beijing Kangmeng Foundation's call to provide medical aid to cancer patients who were impoverished due to illness, and participated in the "Shuxinkeyi-Tumor Immunotherapy Patient Rescue Project" to effectively reduce the financial burden of patients.
(Finish)
Medical Network News on June 7th In China, the incidence and mortality of lung cancer are ranked first.
The reporter learned from Xinda Biotechnology on the 6th that the domestic innovative drug PD-1 inhibitor Daboshu has been approved by the National Medical Products Administration to combine chemotherapy in the first-line treatment of advanced squamous non-small cell lung cancer.
The reporter learned from Xinda Biotechnology on the 6th that the domestic innovative drug PD-1 inhibitor Daboshu has been approved by the National Medical Products Administration to combine chemotherapy in the first-line treatment of advanced squamous non-small cell lung cancer.
Medical experts pointed out that lung cancer is divided into small cell lung cancer and non-small cell lung cancer according to histological type.
Among them, cases of non-small cell lung cancer account for about 80% to 85% of lung cancer patients.
About 30% of Chinese patients with non-small cell lung cancer have squamous non-small cell lung cancer.
Among them, cases of non-small cell lung cancer account for about 80% to 85% of lung cancer patients.
About 30% of Chinese patients with non-small cell lung cancer have squamous non-small cell lung cancer.
Shanghai Pulmonary Medicine Hospital Professor Zhou Caicun oncology director, said the last two decades, progress of drug research and development of non-small cell lung cancer are mainly concentrated in non-squamous non-small cell lung cancer, squamous non-small cell lung cancer because of its unique epidemiology, organization Pathological and molecular biological characteristics, drug development is relatively slow, and patients lack effective drug treatment.
"Tumor, as the number one disease threatening human life and health , has always been a tough topic for researchers and clinicians.
Anti- tumor biologics are also an important research area in our product pipeline.
" said Dr.
Liu Yongjun, President of Cinda Bio, Dabo Shu has a complete layout in the field of squamous non-small cell lung cancer, providing new medication options for lung squamous cell carcinoma patients who previously lacked effective therapeutic drugs.
Anti- tumor biologics are also an important research area in our product pipeline.
" said Dr.
Liu Yongjun, President of Cinda Bio, Dabo Shu has a complete layout in the field of squamous non-small cell lung cancer, providing new medication options for lung squamous cell carcinoma patients who previously lacked effective therapeutic drugs.
It is reported that in Daboshu's layout for squamous non-small cell lung cancer, in addition to the first-line treatment approved this time, the key phase III clinical study of the second-line treatment also achieved positive results.
The listing application was also accepted by the State Drug Administration in January this year .
The listing application was also accepted by the State Drug Administration in January this year .
According to the R&D team, it is currently working hard to expand the scope of indications for Daboshu and has carried out more than 20 clinical studies on Daboshu worldwide.
Clinical research in the fields of colorectal cancer, gastric cancer, cholangiocarcinoma, cervical cancer, lymphoma, and melanoma is also underway.
Clinical research in the fields of colorectal cancer, gastric cancer, cholangiocarcinoma, cervical cancer, lymphoma, and melanoma is also underway.
It is worth mentioning that Daboshu is the first PD-1 in the country to enter the national medical insurance catalog.
In January this year, Cinda Bio responded to the Beijing Kangmeng Foundation's call to provide medical aid to cancer patients who were impoverished due to illness, and participated in the "Shuxinkeyi-Tumor Immunotherapy Patient Rescue Project" to effectively reduce the financial burden of patients.
(Finish)
In January this year, Cinda Bio responded to the Beijing Kangmeng Foundation's call to provide medical aid to cancer patients who were impoverished due to illness, and participated in the "Shuxinkeyi-Tumor Immunotherapy Patient Rescue Project" to effectively reduce the financial burden of patients.
(Finish)
Medical Network News on June 7th In China, the incidence and mortality of lung cancer are ranked first.
The reporter learned from Xinda Biotechnology on the 6th that the domestic innovative drug PD-1 inhibitor Daboshu has been approved by the National Medical Products Administration to combine chemotherapy in the first-line treatment of advanced squamous non-small cell lung cancer.
The reporter learned from Xinda Biotechnology on the 6th that the domestic innovative drug PD-1 inhibitor Daboshu has been approved by the National Medical Products Administration to combine chemotherapy in the first-line treatment of advanced squamous non-small cell lung cancer.
Medical experts pointed out that lung cancer is divided into small cell lung cancer and non-small cell lung cancer according to histological type.
Among them, cases of non-small cell lung cancer account for about 80% to 85% of lung cancer patients.
About 30% of Chinese patients with non-small cell lung cancer have squamous non-small cell lung cancer.
Among them, cases of non-small cell lung cancer account for about 80% to 85% of lung cancer patients.
About 30% of Chinese patients with non-small cell lung cancer have squamous non-small cell lung cancer.
Shanghai Pulmonary Medicine Hospital Professor Zhou Caicun oncology director, said the last two decades, progress of drug research and development of non-small cell lung cancer are mainly concentrated in non-squamous non-small cell lung cancer, squamous non-small cell lung cancer because of its unique epidemiology, organization Pathological and molecular biological characteristics, drug development is relatively slow, and patients lack effective drug treatment.
Hospital hospital hospital "Tumor, as the number one disease threatening human life and health , has always been a tough topic for researchers and clinicians.
Anti- tumor biologics are also an important research area in our product pipeline.
" said Dr.
Liu Yongjun, President of Cinda Bio, Dabo Shu has a complete layout in the field of squamous non-small cell lung cancer, providing new medication options for lung squamous cell carcinoma patients who previously lacked effective therapeutic drugs.
Health Health health diseases disease disease tumor tumor tumorAnti- tumor biologics are also an important research area in our product pipeline.
" said Dr.
Liu Yongjun, President of Cinda Bio, Dabo Shu has a complete layout in the field of squamous non-small cell lung cancer, providing new medication options for lung squamous cell carcinoma patients who previously lacked effective therapeutic drugs.
It is reported that in Daboshu's layout for squamous non-small cell lung cancer, in addition to the first-line treatment approved this time, the key phase III clinical study of the second-line treatment also achieved positive results.
The listing application was also accepted by the State Drug Administration in January this year .
Medicine, medicine, medicineThe listing application was also accepted by the State Drug Administration in January this year .
According to the R&D team, it is currently working hard to expand the scope of indications for Daboshu and has carried out more than 20 clinical studies on Daboshu worldwide.
Clinical research in the fields of colorectal cancer, gastric cancer, cholangiocarcinoma, cervical cancer, lymphoma, and melanoma is also underway.
Clinical research in the fields of colorectal cancer, gastric cancer, cholangiocarcinoma, cervical cancer, lymphoma, and melanoma is also underway.
It is worth mentioning that Daboshu is the first PD-1 in the country to enter the national medical insurance catalog.
In January this year, Cinda Bio responded to the Beijing Kangmeng Foundation's call to provide medical aid to cancer patients who were impoverished due to illness, and participated in the "Shuxinkeyi-Tumor Immunotherapy Patient Rescue Project" to effectively reduce the financial burden of patients.
(Finish)
In January this year, Cinda Bio responded to the Beijing Kangmeng Foundation's call to provide medical aid to cancer patients who were impoverished due to illness, and participated in the "Shuxinkeyi-Tumor Immunotherapy Patient Rescue Project" to effectively reduce the financial burden of patients.
(Finish)