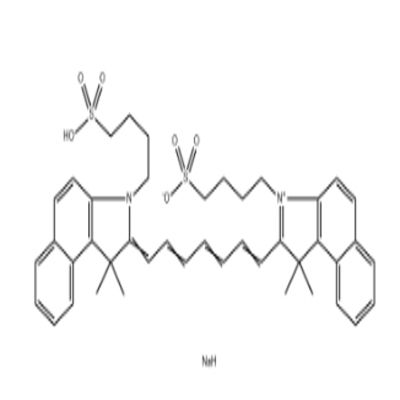
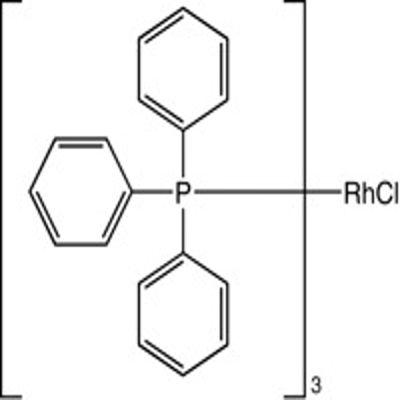
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Pharmaceutical Network Market Analysis In recent years, China has promoted the development of innovative drugs by encouraging innovation and speeding up the reform of the review and approval systemNow China's innovative pharmaceutical industry ushered in an outbreak period, it is expected that by 2020, the number of domestic new drugs clinical Phase I to clinical III varieties will reach a cumulative total of about 1000The number of approved innovative drugs in China is expected to continue to explode from 2021
Since 2015, China has issued a series of policies to encourage pharmaceutical innovation, giving policy support throughout the life cycle of innovative drug approval, clinical trials, production and subsequent marketingSpeed up the approval of drug registration review, clear the approval channel for new drug registration, and reduce waiting times in lineAccording to statistics, the number of CDE annual review and approval of various types of registration applications was about 5000 prior to 2015, and the number of completed registration applications for the whole year 2015-2018 was maintained at the level of 10,000, with the approval capacity doubling significantlyThe accelerated pace of CDE review approval spree has effectively cleared the congestion of the approval channel, and the number of registration applications waiting in line for review has dropped significantly, and in 2018 3,440 registration applications were queued for review approval, down about 85% from the peak of 22,000 in September 2015
In 2019, China adopted the Vaccine Management Law and the Drug Administration Law, which laid an important legal foundation for curing the results of drug review and approval system reform from the legal level, constructing the system of drug whole life cycle supervision and strengthening vaccine supervision. The recent introduction of the Measures for the Administration of Drug Registration is an important operational regulation for drug research and development and registration management in ChinaThe introduction of this series of policies has greatly promoted the development of China's pharmaceutical innovation
2018 is the first year of China's innovation drug industry outbreak, and in 2019 the number of domestic new drugs submitted for listing reached 14, overlaying the accumulation of NDA applications in recent years, the number of domestic new drugs approved in 2020 is expected to increase furtherIt is estimated that by 2020, the number of domestic new drugs clinical Phase I to clinical III varieties will reach a cumulative total of about 1000Considering that the domestic new drugs to me-too-based, the overall success rate is expected to reach about 20%Based on a five-year approval cycle, this means that 100-200 new drugs may be approved in the next 5-10 years, and the number of domestic innovative drugs approved is expected to continue to explode from 2021
In recent years, from the policy environment to industrial support, from capital to talent support, domestic innovation drug research and development continued to accelerate, innovative pharmaceutical enterprises with the advantage of the policy dongfeng and domestic drug structure optimization opportunities, ushered in a golden development periodThe industry said that from a longer cycle, the domestic innovation drugs and their industrial chain has just emerged, this track boom cycle will continue for a long time
However, it is worth noting that the innovation drug research and development cycle is longer, the upfront investment is high, or the sales of innovative drugs are low, some innovative drug companies have long-term losses So what should you think of the valuation of innovative pharmaceutical products or companies? Analysts said that the original valuation approach can no longer simply look at PE or PEG, which focuses on short-term performance growth of one to two years, ignoring the impact of research and development investment on the company's long-term growth The valuation of future innovative drugs will focus on product lines
"But product line valuations are flexible, and each product's sales, each investor's estimate, is different, and there will be a rough hub." Each product is at a different stage and the valuation is different For example, the first phase of clinical new drugs may give a valuation of 200 million, to the third phase of the market will be 2 billion valuation, because the probability of the first phase of product approval is only about 10%, to the third phase of the probability of approval is very high, so even if the same variety, the first phase and the third phase of the valuation is far from As a whole, the varieties under study need to be pulled out, and the three-phase varieties can estimate sales, and then give ps (market-to-market ratio) 3 or 5 times based on sales In addition, also consider the entire company's research and development investment, team professional ability "The above-mentioned people analysis
China's innovative drug market is huge, and given the strong demand from the still-growing domestic drug market and ageing, it could lead to thousands to trillions of incremental markets if the market share is increased from 5% to 50% in the future But innovative drug investment is highly specialized, and risks and challenges coexist.