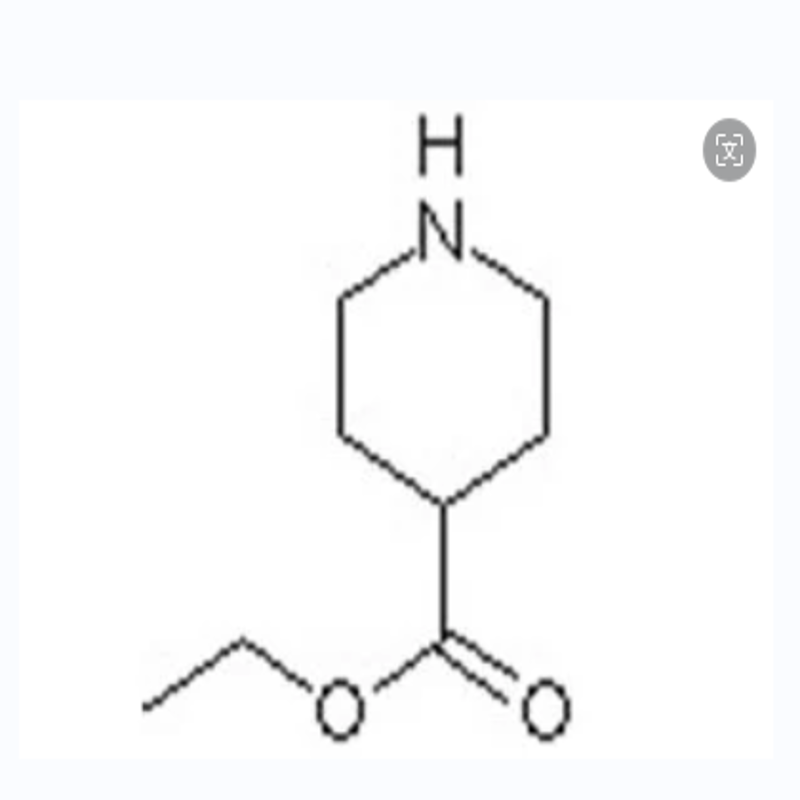
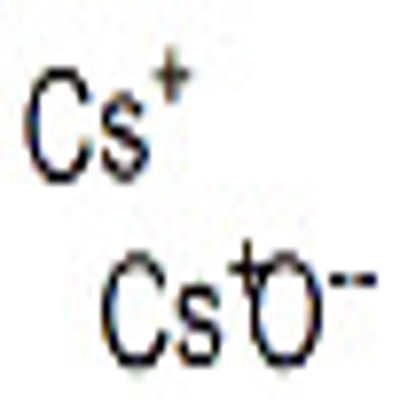-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Recently, the New England Journal of Medicine (NEJM) published the total lifetime and all other secondary endpoint results of the Drarolutamide Phase 3 trial for the treatment of non-metastatic desopathic prostate cancer (nmCRPC).
data showed a significant increase in survival and a 31 per cent reduction in the risk of death in patients treated with drarolutamide.
screenshot Source: New England Journal of Medicine Darolutamide (Nubeqa) is an oral androgen-agent antagonist (ARi) developed by Bayer and Orion to inhibit androgen-like function and the growth of prostate cancer cells.
it was approved by the FDA in July 2019.
applications for listing in China have also been included in the priority review by the Drug Review Center (CDE) of the State Drug Administration of China.
in the trial, called ARAMIS, 1,509 male patients were given a 2:1 group and received aarolutamide (955) or placebo (554) based on androgen deprivation therapy.
previously published results showed that the median non-transferable survival of patients in the darolutamide group was 40.4 months, significantly higher than the 18.4 months in the placebo group, reaching the main study endpoint of the trial.
the main endpoint was confirmed as positive, the trial was blinded and patients in the placebo group were allowed to receive drarolutamide treatment across groups.
At the time of the blinding, all 170 patients who were still receiving placebo treatment switched to drarolutamide; 137 patients who had been deactivated before the test had received at least one other treatment, most commonly dositamine, acetic acid abphetron and encet.
follow-up period was 29.0 months.
the latest data, the three-year total survival rates of the darolutamide group and the placebo group were 83% and 77%, respectively.
risk of death was significantly reduced by 31% compared to the placebo group in the Drarolutamide group.
patients in the Darolutamide group had significantly longer survival times (Photo Source: Reference 1) at other secondary endpoints, darolutamide also showed significant benefits, including the time of the first symptomatic skeletal event and the time of the first use of cytotoxic chemotherapy.
rates of adverse events were similar between the two groups, and no new safety signals were observed.
team concluded that the overall survival benefits of patients in the drarolutamide group were observed even if more than half (55 percent) of patients in the placebo group followed up with drarolutamide or other treatments.
References: ( 1 ) Karim Fizazi, et al., (2020). Nonmetastatic, Castration-Resistant Prostate Cancer and Survival with Darolutamide. N Engl J Med, DOI: 10.1056/NEJMoa2001342 Retrieved September 16, 2020, from