-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Oct 11, 2020 // -- Jazz Pharmaceuticals recently announced positive top-line results for evaluating Xywav (sodium hydroxybutyrate, potassium, magnesium, calcium, JZP-258) oral solution for the treatment of idiopathic hypersomnia (IH) adult patients. prior to
, the FDA granted Xywav fast-track eligibility (FTD) in September 2020 for the treatment of idiopathic narcolepsy (IH), a chronic neurological disorder characterized by excessive drowsiness, uncontrollable sleep demand, or daytime drowsiness for at least three months, even if sleep is adequate or prolonged at night.
was published in a multi-country, double-blind, multi-center, placebo-controlled, randomized drug suspension Phase 3 study that evaluated the efficacy and safety of Xywav in treating adult patients with idiopathic narcolepsy.
the study showed excessive daytime drowsiness, a typical feature of idydic narcolepsy.
study was designed to include a titration and optimization period of up to 14 weeks, a stable dose period of Xywav for 2 weeks, followed by a 1:1 random group of Xywav or placebo treatments for 2 weeks.
patients entered a 24-week open label safety extension period after completing a double-blind, placebo-controlled treatment period.
all patients were treated regularly with Xywav in open-label drops and clinically significant improvements were observed on the Epworth Sleepiness Scale (ESS).
random withdrawal portion of the study involved 115 patients, measuring the overall change in the primary endpoint ESS and key secondary endpoint patients (PGIc) and the Idydlycable Narcolepsy Severity Scale (IHSS).
patients who took Xywav showed clinically significant efficacy in ESS, PGIc, and IHSS, while patients taking placebo showed a highly statistically significant deterioration compared to patients taking placebo: ESS (p-lt;0.0001), PGIc (p-lt;0.0001), IHSS (p-lt;0.0001).
the safety in the study was consistent with the known security of Xywav, and no new safety signals were observed in the population.
Jazz will present data from the phase 3 study at an upcoming medical conference, which will be included in a supplementary new drug application (sNDA) scheduled for the first quarter of 2021.
Dr Robert Iannone, executive vice president of research and development at Jazz, said: "We are excited about these compelling results and the level of improvement observed in the study, particularly the fact that there are currently no approved treatment options for idiophedic narcolepsy groups.
are very grateful to the patients and researchers involved in the study and look forward to working quickly with the FDA to provide Xywav to patients as soon as possible.
15 years, Jazz has been at the forefront of sleep medicine.
our goal is to innovate and change the lives of patients, and we are committed to bringing new options to people with severe sleep disorders who do not have or have limited treatment.
"In July 2020, Xywav was approved by the FDA to treat sudden or excessive daytime drowsiness (EDS) in patients 7 years of age and older with narcolepsy (narcolepsy).
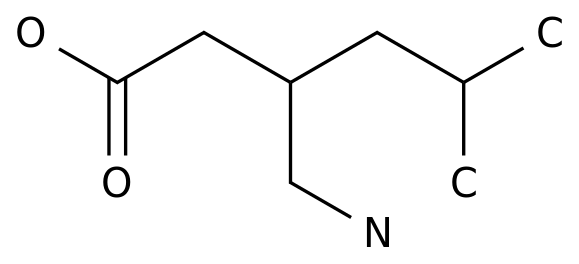
Xywav is a hydroxybutyrate product with a unique cational part (sodium calcium magnesium potassium), which has the same hydroxybutyrate concentration as sodium hydroxybutyrate in the recommended dose range of 6-9 grams, but can reduce sodium by 92%, or about 1000-1500 mg/night.
Although the exact mechanism of Xywav's action is not yet known, Xywav's therapeutic effects on palladiosis and EDS are thought to be mediated by the GABAB effects on epinephrine and dopamine neurons and hypothyroid neurons during sleep.
Sodium hydroxybutyrate, which contains high sodium levels, was previously the only approved product for the treatment of edding and EDS in patients 7 years of age and older with narcissitis, and has been designated by the American Academy of Sleep Medicine (AASM) as the standard of care for the treatment of sudden onset and EDS.
narcolepsy (narcolepsy) is an incurable chronic neurological disease that can have a profound impact on a patient's health over time.
many patients may go through years before they get a correct diagnosis, which can have a significant impact on their daily lives.
is a lifelong disease, so it is important to have new options to help treat EDS and collapse.
Xywav was developed specifically to provide low-sodium hydroxybutyric acid therapy for patients with densophpathy, and without warning of sodium levels, the drug will become a new standard of care.
Xywav is available in a variety of dosage options for adults and children.
prescribers can dipped Xywav into different doses for patients to take at night.
When a patient is switched from sodium hydroxybutyrate to Xywav treatment, Xywav's starting treatment dose and program are the same as sodium hydroxybutyrate (by grams) and can be titrated on demand according to efficacy and tolerance.
Origin: Jazz Pharmaceuticals Announces Positive Top-line Results from Phase 3 Study of Xywav? (calcium, magnesium, potassium, and sodium oxybates) Oral Solutions in Adult Patients with Idiopathic Hypersomnia.