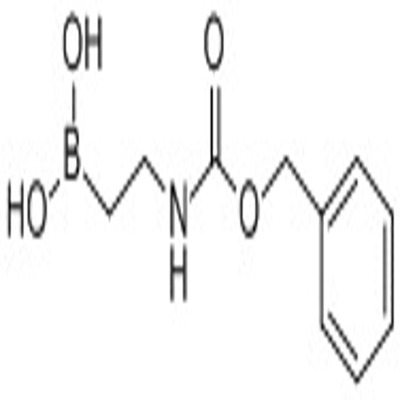
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
(2020/09/15) Sanofi/regenerative Dupixent treatment EoE was recognized by the FDA breakthrough therapy; "market exclusive period" one-year patent link waiting period reduced to 9 months to innovate or first imitation; Zhong Nanshan team: human recombinant granulocyte set stimulation factor to reduce the death of specific new crown patients ... (Click on the title, available in the original article) Sanofi/Regenerative Dupixent Treatment EoE was recognized by the FDA Breakthrough Therapy on September 14th, and the FDA has awarded Sanofi/Regenerative Dupixent (dupilumab, Dupilumab) breakthrough treatment designation for the treatment of patients aged 12 years and older with eosinophilic esophonic esopheditis (EoE).
as early as 2017, Dupixent was also awarded the title of orphan drug for potential EoE treatment.
: Dupixent has conducted 50 clinical trials in more than 10,000 patients.
:Pharmaceutical Rubik's Cube Gilead: On September 14th Gilead Sciences held an investor meeting on a $21 billion cash acquisition of Immunomedics, revealing that Gilead had crafted a number of deals with 13 outside innovative pharmaceutical companies in the past two years, involving a total of more than $40 billion in deals aimed at improving its internal innovation capabilities.
headlines: you can only lament "money."
"E drug manager" "market exclusive period" one-year patent link waiting period reduced to 9 months to innovate or first imitation recently, the State Drug Administration issued the "drug patent dispute early settlement mechanism implementation measures (trial)" for comments.
The draft opinion, which proposes a "waiting period" for first-generic companies to apply for patent challenges when approved by the Drug Administration, and a "market exclusive period" for first-generic drugs, is considered to be the domestic version of the Hatch-Waxman Act (the U.S. Drug Price Competition and Patent Term Amendment), and these two provisions are also considered to be clearly good for first-generic companies.
the headline bacteria: low-growth peaches are picked up.
New Medical View point of view: Human recombinant granulocyte collection stimulation factor to reduce the death of specific new coronary patients recently, Zhong Nanshan academician team in the Journal of the American Medical Association - Internal Medicine published the human recombinant granulocyte collection stimulation factor (rhG-CSF) in the new coronavirus disease (COVID-19) accompanied by lymphocyte reduction but no merger of patients in a randomized controlled clinical trial results.
showed that rhG-CSF may have reduced the number of patients with severe illness or death in these patients.
: Similar time to clinical improvement between the two groups.
the "Mi Net" exclusive injection surge 400% biopharmaceous harvest! Lizhu half a year to earn 1 billion September 10, Lizhu Group announced that the company after 5 years of research and development of injection of octreotide acetate micro-ball approved clinical, the product is a long-lasting slow release preparation, currently only import approval, the company's cumulative investment in research and development costs of about 26.96 million yuan.
In the first half of 2020, Lizhu Group's exclusive product injection with acetate bright propylene rhapsody micro-ball achieved revenue of 536 million yuan, an increase of 17.26%, the company's micro-ball platform in the research of new products with acetate querin micro-ball, injection with acetaminophen micro-ball is expected to be listed after 2022, as a new boost to performance.
: The outbreak led to a surge in reagents.
, a well-known pharmaceutical company to buy six large varieties recently, Fangsheng Pharmaceutical announced that Fangsheng Pharmaceuticals and Hunan Kangjia Pharmaceutical signed a "drug variety transfer agreement." according to the
announcement, Fangsheng Pharmaceuticals decided to transfer the ownership of six drug varieties, such as "intestinal capsules", "vitamin C bubble particles", "through pulse oral solution", "six-flavored yellow capsules", "silver yellow particles" and "A gum sanbao paste" for RMB7 million.
: The sale of the subsidiary was unsuccessful.




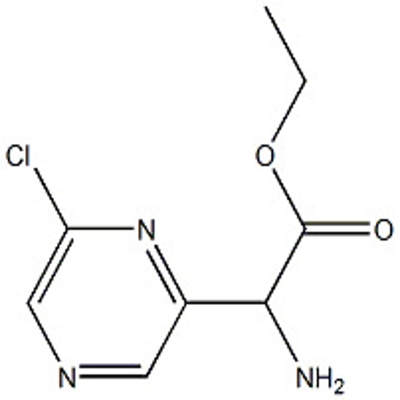
![[4-methylsulfonyl-2-(trifluoromethyl)phenyl]boronic acid](https://file.echemi.com/fileManage/upload/goodpicture/20210823/m20210823171652704.jpg)