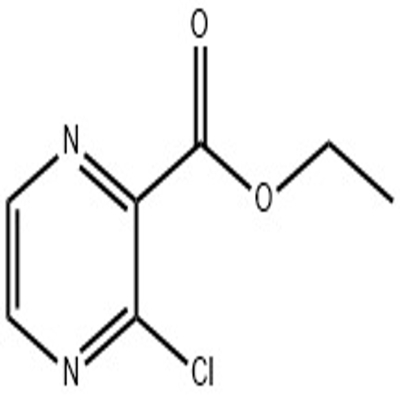
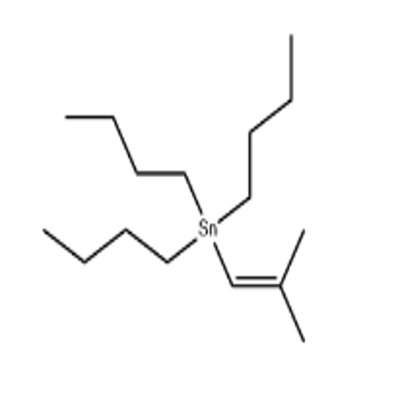
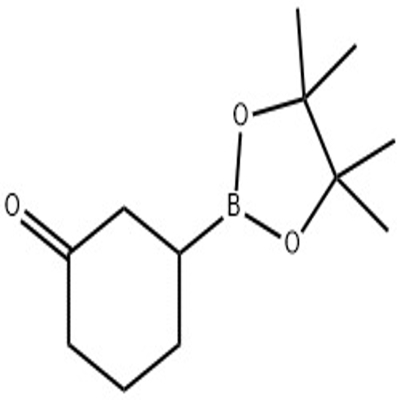
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
From April 26th to April 21st, 2021, the listing application of VERKAZIA (0.
VERKAZIA (0.
Santian VERKAZIA (0.
On November 19, 2020, Santian VERKAZIA (0.
The “List of New Drugs Urgently Needed in Clinics (Third Batch)” is a clinical trial selected after preliminary screening by the National Drug Evaluation Center, and the National Medical Products Administration and the National Health and Health Commission jointly organizing clinical experts to conduct research and demonstration of clinically urgently needed overseas new drugs.
For varieties that are included in the list of urgently needed overseas new drugs for clinical use, relevant materials can be submitted in accordance with the ``Working Procedures for the Review and Approval of New Drugs Urgently Needed in Clinics'', and they can directly submit a listing application to the Drug Evaluation Center of the National Medical Products Administration, and enter the fast track for review and approval.
Recently, the listing application of VERKAZIA (0.
Verkazia (0.
Breakthrough treatment difficulties spring keratoconjunctivitis has attracted much attention
Vernal keratoconjunctivitis (VKC) is a common chronic allergic conjunctivitis, also known as "spring catarrhal conjunctivitis".
According to the ``Expert Consensus on the Diagnosis and Treatment of Allergic Conjunctivitis in China (2018)'' (hereinafter referred to as the ``Consensus''), 10%-40% of the world's population has a history of allergic conjunctivitis, of which VKC is a major influence Severe allergic ocular surface disease of the conjunctiva and cornea
For patients with severe refractory VKC, long-term medication is needed.
At the 2021 CCCRS conference held not long ago, in the "VKC" diagnosis and treatment seminar held by Santian Company, relevant ophthalmologists stated that the pathogenesis of VKC involves two important pathways.
Studies have shown that 0.
A randomized, double-blind, critical clinical study evaluating the efficacy and safety of VERKAZIA (0.
Looking forward to the future listing of Verkazia®, it will provide ophthalmologists with a more effective and safer choice in the VKC management program, and bring the gospel to Chinese VKC patients