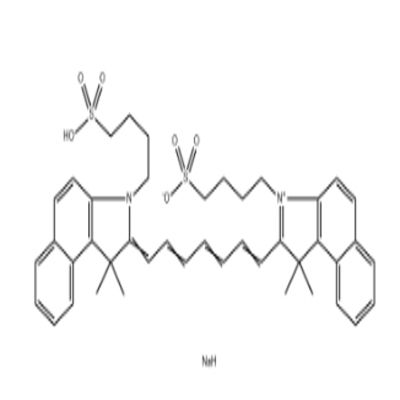
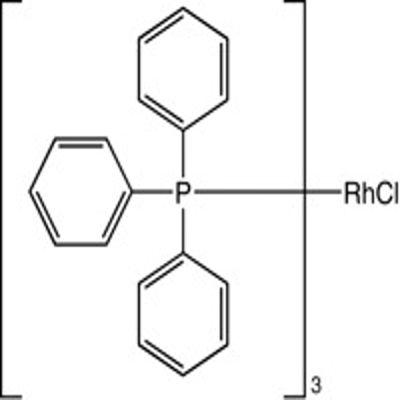
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Health and Family Planning Commission of provinces, autonomous regions and municipalities directly under the Central Government, Xinjiang Production and Construction Corps Health Bureau:
In order to further strengthen post-mortem supervision in the clinical application of medical technology, do a good job in the clinical application management of "restricting clinical application" medical technology, standardize clinical behavior, ensure medical quality and medical safety, I Commission The tissue system (repair) has formulated 15 medical technology management norms for "restricting clinical application" such as the Hematopoietic Stem Cell Transplant Technology Management Code (2017 edition), and has formulated quality control indicators for the corresponding technologies (available for download from the website of the National Health and Family Planning Commission). It is now issued to you, please comply with the implementation.
The Technical Management Code for Transgender Surgery (Trial) issued on 13 November 2009 (Health Care Administration (2009) No. 185), and the Technical Management Code for the Application of Cell Assistive Devices (Trial) (2009) No. 186), "Technical Management Practices for Radioactive Particle Implantation Treatment (Trial)" (Health Care Administration (2009) No. 187), "Technical Management Practices for Deep Tumor Therapy and Systemic Thermal Therapy (Trial)" (Health Care Medical Administration (2009) 188) No.), "Technical Management Practices for Hematopoietic Stem Cell Therapy (Trial)" (SSE Medical Administration No. 189), "Technical Management Code for Tumor Ablation Treatment (Trial)" (SSE Hematopoietic Stem Cell Treatment 190), Oral Jaws Technical Management Specifications for Joint Rooting Technology of Tumors and Jaws (Trial) (Health Care Administration (2009) No. 191), "Technical Management Practice for Craniofacial Malformation of Craniofacial Surgery (Trial)" (Wei's Medical Administration (2009) No. 192), " Technical Management Specifications for the Implantation of Radioactive Particles for Therapy of Malignant Tumors in the Mouth Jaws (Trial) (SSE Medical Administration (2009) No. 193), Andy Yan Face Allogeneic Organ Transplantation Technology Management Code (Trial) (Wei-SE Medical Administration (2009) No. 193) 2009 No. 194), "Genetic Chip Diagnostic Technology Management Code (Trial)" (Wei-Do Medical Administration (2009) No.195), "Artificial Intelligence-assisted Diagnostic Technology Management Code (Trial)" (Wei-Run Medical Administration (2009) No. 196), Artificial Intelligence Assisted Treatment Technology Management Code (Trial) (Wei-SE Medical Administration (2009) No. 197), Proton and Heavy Ion Accelerator Radiotherapy Technology Management Code (Trial) (Health Care Administration (2009) No. 198), "Organization engineering tissue transplant treatment technical management norms (trial)" (Health Care Administration (2009) No. 199) shall be abolished at the same time. (Health and Planning Commission)