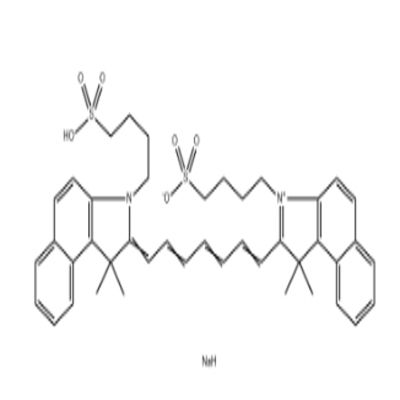
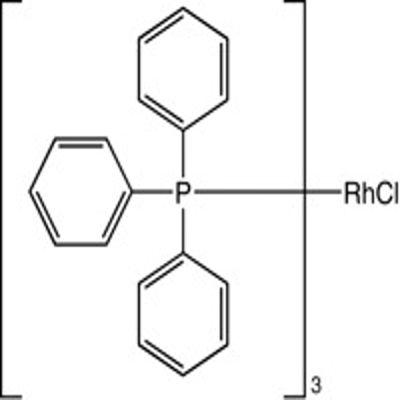
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Medical Network, June 29, in order to fully implement the "Implementation Opinions of the General Office of the State Council on Comprehensively Strengthening the Construction of Drug Regulatory Capability" (Guobanfa [2021] No.
16), the State Food and Drug Administration is comprehensively summarizing China's drug regulatory scientific actions Based on the implementation of the first batch of key projects planned, the second batch of 10 key projects was determined and released
.
The 10 key projects are the effectiveness and safety evaluation of traditional Chinese medicine and the whole process quality control research, stem cell and gene therapy product evaluation system and method research, real-world data support traditional Chinese medicine, rare disease treatment drugs, innovation and clinical urgently needed medical device evaluation methods research , Evaluation research on the diagnosis and treatment of emerging infectious diseases, research on the safety, effectiveness and quality control evaluation of nano-innovative drugs, medical devices, evaluation research on innovative medical devices based on remote transmission, flexible electronic technology and medical robots, new biological Research on material safety and effectiveness evaluation, research on technical guidelines for new cosmetic raw materials, research on cosmetic safety monitoring and analysis early warning methods, research on new tools, new standards and new methods for evaluation of common diseases and frequently-occurring diagnosis and treatment products such as malignant tumors, and drug and medical device vigilance Technology and method research
.
In principle, the execution cycle of this batch of key projects is 2 years
.
Each project is led by the relevant departments and bureaus of the State Food and Drug Administration, and implemented by relevant directly affiliated units.
In principle, the cooperative units rely on the State Food and Drug Administration to supervise scientific research bases and key laboratories
.
The State Food and Drug Administration requires all leading units and implementing units to research and formulate project implementation plans, clarify research plans, refine research objectives and tasks, implement cooperative units, and accelerate innovation in accordance with the principles of focusing on frontiers, highlighting key points, strengthening actual results, and steadily advancing.
Regulatory tools, standards, and methods will further enhance drug regulatory capabilities and levels, accelerate the launch of innovative products, and better meet public health needs
.
The China Pharmaceutical Regulatory Scientific Action Plan was launched in April 2019, and the first batch of 9 key research projects were simultaneously identified
.
After two years of hard work, the first batch of key projects has achieved important results, and 103 new regulatory tools, new methods, and new standards have been studied and formulated , 31 of which have been released
.
16), the State Food and Drug Administration is comprehensively summarizing China's drug regulatory scientific actions Based on the implementation of the first batch of key projects planned, the second batch of 10 key projects was determined and released
.
The 10 key projects are the effectiveness and safety evaluation of traditional Chinese medicine and the whole process quality control research, stem cell and gene therapy product evaluation system and method research, real-world data support traditional Chinese medicine, rare disease treatment drugs, innovation and clinical urgently needed medical device evaluation methods research , Evaluation research on the diagnosis and treatment of emerging infectious diseases, research on the safety, effectiveness and quality control evaluation of nano-innovative drugs, medical devices, evaluation research on innovative medical devices based on remote transmission, flexible electronic technology and medical robots, new biological Research on material safety and effectiveness evaluation, research on technical guidelines for new cosmetic raw materials, research on cosmetic safety monitoring and analysis early warning methods, research on new tools, new standards and new methods for evaluation of common diseases and frequently-occurring diagnosis and treatment products such as malignant tumors, and drug and medical device vigilance Technology and method research
.
In principle, the execution cycle of this batch of key projects is 2 years
.
Each project is led by the relevant departments and bureaus of the State Food and Drug Administration, and implemented by relevant directly affiliated units.
In principle, the cooperative units rely on the State Food and Drug Administration to supervise scientific research bases and key laboratories
.
The State Food and Drug Administration requires all leading units and implementing units to research and formulate project implementation plans, clarify research plans, refine research objectives and tasks, implement cooperative units, and accelerate innovation in accordance with the principles of focusing on frontiers, highlighting key points, strengthening actual results, and steadily advancing.
Regulatory tools, standards, and methods will further enhance drug regulatory capabilities and levels, accelerate the launch of innovative products, and better meet public health needs
.
The China Pharmaceutical Regulatory Scientific Action Plan was launched in April 2019, and the first batch of 9 key research projects were simultaneously identified
.
After two years of hard work, the first batch of key projects has achieved important results, and 103 new regulatory tools, new methods, and new standards have been studied and formulated , 31 of which have been released
.