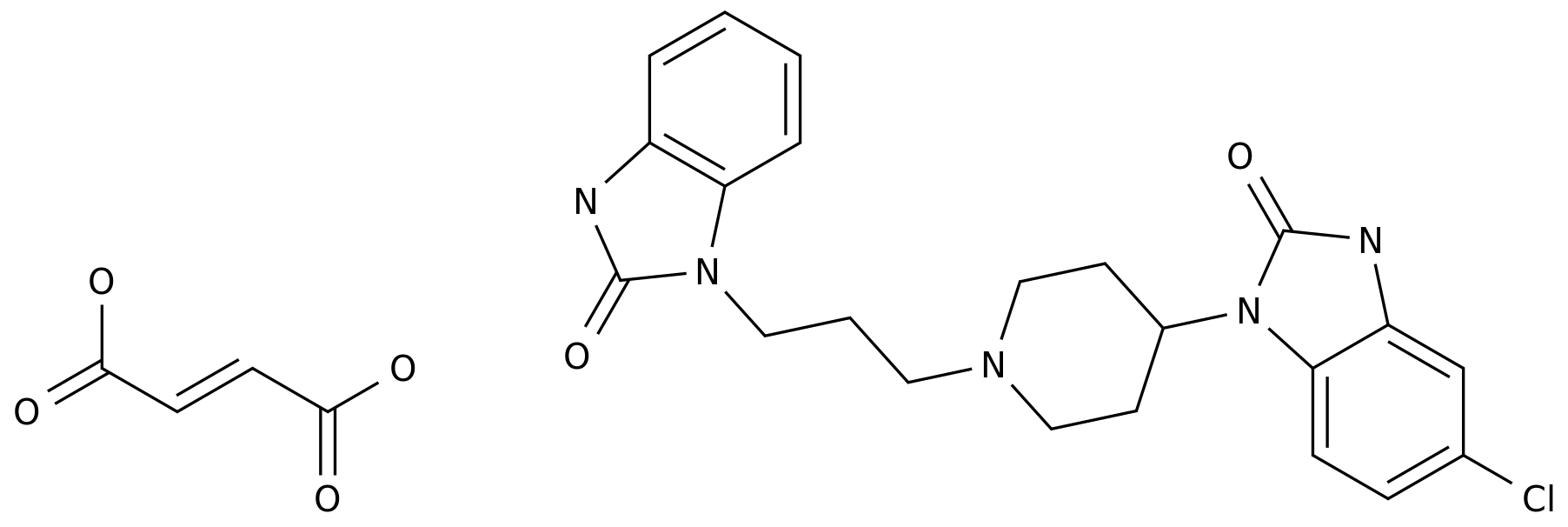
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
- Cosmetic Ingredient
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Editor’s note iNature is China’s largest academic public account.
It is jointly created by a team of doctors from Tsinghua University, Harvard University, Chinese Academy of Sciences and other units.
The iNature talent public account is now launched, focusing on talent recruitment, academic progress, scientific research information, interested parties can Long press or scan the QR code below to follow us
.
iNature drug-induced liver injury (DILI) has been recognized as an important cause of acute and chronic liver disease (CLD)
.
Although most patients who experience DILI recover biochemically and liver enzymes return to normal after stopping suspicious drugs, 8% to 20% of patients will progress to chronic DILI
.
So far, the diagnosis of chronic DILI has become a challenge in liver diseases due to the complex clinical manifestations and lack of specific biomarkers
.
In addition, the relationship between histological characteristics and biochemical characteristics and prognosis of DILI is still unclear
.
On December 16, 2021, Ji Dong, Zou Zhengsheng and Xiao Xiao-He from the Fifth Medical Center of the PLA General Hospital published an online publication titled “Prediction of Biochemical Nonresolution in Patients with Chronic Drug-induced Liver Injury” in Hepatology (IF=17) : A Large Multicenter Study" research paper, the purpose of this study is to clarify the high-risk factors of chronic drug-induced liver injury (DILI) and develop a new nomogram model to predict the biochemistry of patients with chronic drug-induced liver injury (DILI) Resolution (BR) or not (BNR)
.
The study was a retrospective study.
3,655 of the 5,326 chronic DILI patients came from 9 participating hospitals, of which 2,866 received liver biopsy
.
All these patients have been followed for more than a year, and their clinical characteristics were retrieved from the electronic medical records
.
The endpoint is BNR, defined as alanine aminotransferase (ALT) or aspartate aminotransferase (AST)> 1.
5 × upper limit of normal (ULN) or alkaline phosphatase (ALP)> 1.
1 × ULN, 12 after the diagnosis of chronic DILI Month
.
The non-invasive high-risk factors of BNR determined by multivariate logistic regression were used to establish a nomogram and verified in an independent external cohort
.
Finally, 19.
3% (707/3,655) of patients had BNR
.
Histologically, with the increase of liver inflammation grade and fibrosis stage, the proportion of BNR increased significantly
.
Compared with no inflammation or mild inflammation, the risk of BNR in patients with obvious inflammation increased by 21.
3 times (P <0.
001)
.
In terms of biochemistry, AST and total bilirubin, platelets, prothrombin time, gender and age are related to BNR, and are included in the construction of a nomogram model (BNR-6), with a C index of 0.
824 (95% CI, 0.
798-0.
849) ), which is highly consistent with liver histology
.
These results have been successfully verified in both the internal queue and the external queue
.
In summary, the study found that significant liver inflammation is a powerful predictor of biochemical failure
.
The established BNR-6 model provides an easy-to-use method to evaluate the outcome of chronic DILI
.
In recent years, drug-induced liver injury (DILI) has been recognized as an important cause of acute and chronic liver disease (CLD)
.
In China, the problem of DILI is particularly prominent due to the random use of Chinese medicines (TCMs) or herbal and dietary supplements (HDS)
.
Although most patients who experience DILI recover biochemically and liver enzymes return to normal after discontinuing suspected drugs, 8% to 20% of patients will progress to chronic DILI, which is characterized by persistent abnormalities in liver examination and histological or radiological evidence , Continuous liver damage for more than 6 months
.
Patients with consecutive episodes of liver damage may develop cirrhosis or even liver failure, as well as a worse quality of life
.
The severe consequences of chronic DILI may result in lost working hours and increased health care expenditures
.
Articles currently published in DILI mostly focus on the investigation of the incidence of DILI, the types of suspected drugs, age distribution, clinical features, pathogenesis, and the establishment of animal models
.
Although early prediction and risk assessment may be the future direction, some previous studies have shown that some factors such as women, older age, clinical patterns, total bilirubin (TBIL) and high inflammation may increase the adverse effects of DILI results Risk
.
So far, the diagnosis of chronic DILI has become a challenge in liver diseases due to the complex clinical manifestations and lack of specific biomarkers
.
Liver biopsy helps to diagnose and differentiate DILI
.
However, it is considered limited due to its invasive methods and high cost
.
In addition, the relationship between histological characteristics and biochemical characteristics and prognosis of DILI is still unclear
.
The purpose of this study is to clarify the high risk factors of chronic drug-induced liver injury (DILI) and develop a new nomogram model to predict the biochemical resolution (BR) or not (BNR) of patients with chronic drug-induced liver injury (DILI)
.
The study was a retrospective study.
3,655 of the 5,326 chronic DILI patients came from 9 participating hospitals, of which 2,866 received liver biopsy
.
All these patients have been followed for more than a year, and their clinical characteristics were retrieved from the electronic medical records
.
The endpoint is BNR, defined as alanine aminotransferase (ALT) or aspartate aminotransferase (AST)> 1.
5 × upper limit of normal (ULN) or alkaline phosphatase (ALP)> 1.
1 × ULN, 12 after the diagnosis of chronic DILI Month
.
The non-invasive high-risk factors of BNR determined by multivariate logistic regression were used to establish a nomogram and verified in an independent external cohort
.
Finally, 19.
3% (707/3,655) of patients had BNR
.
Histologically, with the increase of liver inflammation grade and fibrosis stage, the proportion of BNR increased significantly
.
Compared with no inflammation or mild inflammation, the risk of BNR in patients with obvious inflammation increased by 21.
3 times (P <0.
001)
.
In terms of biochemistry, AST and total bilirubin, platelets, prothrombin time, gender and age are related to BNR, and are included in the construction of a nomogram model (BNR-6), with a C index of 0.
824 (95% CI, 0.
798-0.
849) ), which is highly consistent with liver histology
.
These results have been successfully verified in both the internal queue and the external queue
.
In summary, the study found that significant liver inflammation is a powerful predictor of biochemical failure
.
The established BNR-6 model provides an easy-to-use method to evaluate the outcome of chronic DILI
.
Reference message: https://aasldpubs.
onlinelibrary.
wiley.
com/doi/10.
1002/hep.
32283
It is jointly created by a team of doctors from Tsinghua University, Harvard University, Chinese Academy of Sciences and other units.
The iNature talent public account is now launched, focusing on talent recruitment, academic progress, scientific research information, interested parties can Long press or scan the QR code below to follow us
.
iNature drug-induced liver injury (DILI) has been recognized as an important cause of acute and chronic liver disease (CLD)
.
Although most patients who experience DILI recover biochemically and liver enzymes return to normal after stopping suspicious drugs, 8% to 20% of patients will progress to chronic DILI
.
So far, the diagnosis of chronic DILI has become a challenge in liver diseases due to the complex clinical manifestations and lack of specific biomarkers
.
In addition, the relationship between histological characteristics and biochemical characteristics and prognosis of DILI is still unclear
.
On December 16, 2021, Ji Dong, Zou Zhengsheng and Xiao Xiao-He from the Fifth Medical Center of the PLA General Hospital published an online publication titled “Prediction of Biochemical Nonresolution in Patients with Chronic Drug-induced Liver Injury” in Hepatology (IF=17) : A Large Multicenter Study" research paper, the purpose of this study is to clarify the high-risk factors of chronic drug-induced liver injury (DILI) and develop a new nomogram model to predict the biochemistry of patients with chronic drug-induced liver injury (DILI) Resolution (BR) or not (BNR)
.
The study was a retrospective study.
3,655 of the 5,326 chronic DILI patients came from 9 participating hospitals, of which 2,866 received liver biopsy
.
All these patients have been followed for more than a year, and their clinical characteristics were retrieved from the electronic medical records
.
The endpoint is BNR, defined as alanine aminotransferase (ALT) or aspartate aminotransferase (AST)> 1.
5 × upper limit of normal (ULN) or alkaline phosphatase (ALP)> 1.
1 × ULN, 12 after the diagnosis of chronic DILI Month
.
The non-invasive high-risk factors of BNR determined by multivariate logistic regression were used to establish a nomogram and verified in an independent external cohort
.
Finally, 19.
3% (707/3,655) of patients had BNR
.
Histologically, with the increase of liver inflammation grade and fibrosis stage, the proportion of BNR increased significantly
.
Compared with no inflammation or mild inflammation, the risk of BNR in patients with obvious inflammation increased by 21.
3 times (P <0.
001)
.
In terms of biochemistry, AST and total bilirubin, platelets, prothrombin time, gender and age are related to BNR, and are included in the construction of a nomogram model (BNR-6), with a C index of 0.
824 (95% CI, 0.
798-0.
849) ), which is highly consistent with liver histology
.
These results have been successfully verified in both the internal queue and the external queue
.
In summary, the study found that significant liver inflammation is a powerful predictor of biochemical failure
.
The established BNR-6 model provides an easy-to-use method to evaluate the outcome of chronic DILI
.
In recent years, drug-induced liver injury (DILI) has been recognized as an important cause of acute and chronic liver disease (CLD)
.
In China, the problem of DILI is particularly prominent due to the random use of Chinese medicines (TCMs) or herbal and dietary supplements (HDS)
.
Although most patients who experience DILI recover biochemically and liver enzymes return to normal after discontinuing suspected drugs, 8% to 20% of patients will progress to chronic DILI, which is characterized by persistent abnormalities in liver examination and histological or radiological evidence , Continuous liver damage for more than 6 months
.
Patients with consecutive episodes of liver damage may develop cirrhosis or even liver failure, as well as a worse quality of life
.
The severe consequences of chronic DILI may result in lost working hours and increased health care expenditures
.
Articles currently published in DILI mostly focus on the investigation of the incidence of DILI, the types of suspected drugs, age distribution, clinical features, pathogenesis, and the establishment of animal models
.
Although early prediction and risk assessment may be the future direction, some previous studies have shown that some factors such as women, older age, clinical patterns, total bilirubin (TBIL) and high inflammation may increase the adverse effects of DILI results Risk
.
So far, the diagnosis of chronic DILI has become a challenge in liver diseases due to the complex clinical manifestations and lack of specific biomarkers
.
Liver biopsy helps to diagnose and differentiate DILI
.
However, it is considered limited due to its invasive methods and high cost
.
In addition, the relationship between histological characteristics and biochemical characteristics and prognosis of DILI is still unclear
.
The purpose of this study is to clarify the high risk factors of chronic drug-induced liver injury (DILI) and develop a new nomogram model to predict the biochemical resolution (BR) or not (BNR) of patients with chronic drug-induced liver injury (DILI)
.
The study was a retrospective study.
3,655 of the 5,326 chronic DILI patients came from 9 participating hospitals, of which 2,866 received liver biopsy
.
All these patients have been followed for more than a year, and their clinical characteristics were retrieved from the electronic medical records
.
The endpoint is BNR, defined as alanine aminotransferase (ALT) or aspartate aminotransferase (AST)> 1.
5 × upper limit of normal (ULN) or alkaline phosphatase (ALP)> 1.
1 × ULN, 12 after the diagnosis of chronic DILI Month
.
The non-invasive high-risk factors of BNR determined by multivariate logistic regression were used to establish a nomogram and verified in an independent external cohort
.
Finally, 19.
3% (707/3,655) of patients had BNR
.
Histologically, with the increase of liver inflammation grade and fibrosis stage, the proportion of BNR increased significantly
.
Compared with no inflammation or mild inflammation, the risk of BNR in patients with obvious inflammation increased by 21.
3 times (P <0.
001)
.
In terms of biochemistry, AST and total bilirubin, platelets, prothrombin time, gender and age are related to BNR, and are included in the construction of a nomogram model (BNR-6), with a C index of 0.
824 (95% CI, 0.
798-0.
849) ), which is highly consistent with liver histology
.
These results have been successfully verified in both the internal queue and the external queue
.
In summary, the study found that significant liver inflammation is a powerful predictor of biochemical failure
.
The established BNR-6 model provides an easy-to-use method to evaluate the outcome of chronic DILI
.
Reference message: https://aasldpubs.
onlinelibrary.
wiley.
com/doi/10.
1002/hep.
32283