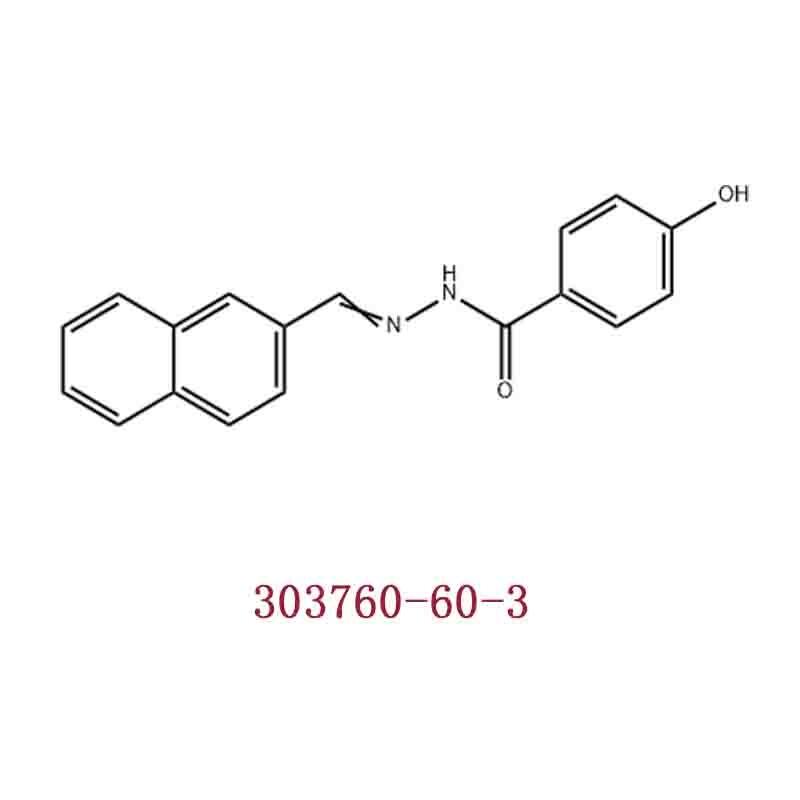
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
iNature
Despite neoadjuvant/translational chemotherapy, cT4a/bN+ has a poor
prognosis for gastric cancer.
Immune checkpoint inhibitors (ICIs) and antiangiogenic drugs have shown activity in advanced gastric cancer, but their efficacy in neoadjuvant/translational settings is unclear
.
On January 3, 2023, the team of Liu Lian of Shandong University published a report entitled "Neoadjuvant therapy with immune checkpoint blockade, antiangiogenesis, and chemotherapy for locally advanced gastric" online in Nature Communications cancer", which conducted a single-arm phase II exploratory trial evaluating the efficacy
of ICI (camrelizumab), antiangiogenic (apatinib), and chemotherapy (S-1±oxaliplatin) in combination with cT4a/bN+ neoadjuvanc/conversion therapy for gastric cancer.
The results showed that the complete and major pathological response rates were 15.
8% and 26.
3%,
respectively.
The pathological response was significantly correlated
with microsatellite instability, PD-L1 expression and tumor mutational burden.
In addition, multiomics revealed several presumptive biomarkers of pathological responses, including RREB1 and SSPO mutations, immune-relevant features, and peripheral T cell expansion scores
.
Multiomics also demonstrated dynamic changes
in dominant tumor subclonals, immune microenvironment, and T cell receptor repertoire during neoadjuvant immunotherapy.
Toxicity and postoperative complications are limited
.
These data support further validation of ICI- and antiangiogenesis-based neoadjuvant/conversion therapies in large randomized trials and provide candidate biomarkers
.
in mortality.
Radical resection is the best option
for treatment and prolonging survival.
In previous MAGIC and FFCD clinical trials, perioperative chemotherapy had a higher 5-year survival rate than surgery
alone.
The FLOT regimen further improved R0 resection rates and prolonged overall survival (OS)
compared with the no paclitaxel regimen.
However, cT4 patients accounted for less than 10%
of patients in this trial.
In a recent PRODIGY study, neoadjuvant DOS regimens (docetaxel, oxaliplatin, and S-1 [Tegafur/Gimestat/Oxonate]) and adjuvant S-1 significantly improved 3-year progression-free survival (PFS) (66.
3 versus 59.
8 months) for postoperative S-1 and resulted in a 10.
4% complete pathologic response
.
Although 70% of patients in this trial had cT4, a whopping 89%
of patients with cT4a were present.
In addition, the prognosis was lower in the cT4N+ group than in the cT4N- and cT2-3N+ groups, and there was no separate subgroup analysis
for cT4bN+ patients.
Recently, in phase III randomized controlled trials, immune checkpoint inhibitors (ICIs) achieved superior outcomes
over placebo in third-line treatment of advanced gastric cancer.
In first-line therapy, in patients with PD-L1 CPS≥5 in CheckMate 6498 and ORIENT169, combining ICI and chemotherapy prolongs OS and PFS and reduces the risk of death by 20-35%
compared with chemotherapy alone.
Theoretically, due to the intact immune system, sufficient neoantigens, and low tumor clonality, the neoadjuvant therapeutic environment is the best choice
for immunotherapy.
ICI-based neoadjuvant therapy has been successfully used for resectable non-small cell lung cancer
.
In gastric cancer, especially in locally advanced gastric cancer, the role of chemotherapy remains incompletely explored
.
Genomic characteristics and clonal evolution after neoadjuvant therapy (Figure from Nature Communications) Tumor angiogenesis plays an important role
in tumor development.
Like ICIs, antiangiogenic drugs target tumor microenvironment (TME) components rather than tumor cells and work
synergistically with ICIs by promoting CD8+ T lymphocyte infiltration and activation.
Ramucirumab (an anti-VEGFR2 antibody) and apatinib (a VEGFR2 tyrosine kinase inhibitor) have been shown to prolong OS and are approved for second- and third-line treatment
of advanced gastric cancer, respectively.
In some phase I/II studies, they have been shown to reprogram TME, reverse immunosuppression to inflammatory states, and enhance the efficacy
of ICIs.
Therefore, the addition of antiangiogenic drugs to ICIs plus chemotherapy regimens may enhance the efficacy
of neoadjuvant therapy.
In this phase II trial, the authors' data suggest that ICI-based and antiangiogenesis-based neoadjuvant/conversion therapies have good efficacy and feasibility
in cT4a/bN+ gastric cancer, particularly in MSI-H and PD-L1-positive patients.
How to improve its efficacy in MSS and PD-L1 negative patients needs to be further explored
.
The results of the multiomics study provide a number of candidate biomarkers associated with efficacy and help understand the mechanisms
of treatment response and drug resistance.
Original link: style="margin-right: auto;margin-left: auto;outline: 0px;width: 30px;display: inline-block;">
—END—
The content is [iNature]