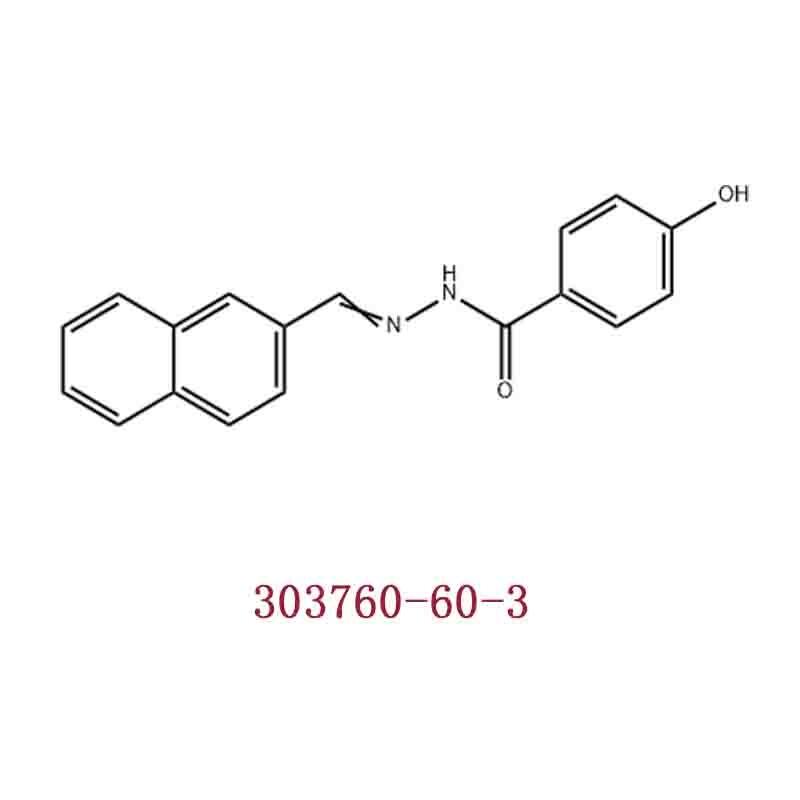
-
Categories
-
Pharmaceutical Intermediates
-
Active Pharmaceutical Ingredients
-
Food Additives
- Industrial Coatings
- Agrochemicals
- Dyes and Pigments
- Surfactant
- Flavors and Fragrances
- Chemical Reagents
- Catalyst and Auxiliary
- Natural Products
- Inorganic Chemistry
-
Organic Chemistry
-
Biochemical Engineering
- Analytical Chemistry
-
Cosmetic Ingredient
- Water Treatment Chemical
-
Pharmaceutical Intermediates
Promotion
ECHEMI Mall
Wholesale
Weekly Price
Exhibition
News
-
Trade Service
Only for medical professionals to read for reference.
The 9th Asian Inflammatory Bowel Disease Conference and China Inflammatory Bowel Disease Annual Conference (AOCC) will be held on October 14-16, 2021 in Guangzhou, China
.
Crohn's disease (CD) is a type of inflammatory bowel disease (IBD), which can occur in any part of the gastrointestinal tract.
It is a chronic, recurrent and non-specific gastrointestinal disease.
The cause of transmural inflammation is unclear so far [1]
.
Half of the patients required surgery within the first 10 years and the risk of postoperative recurrence reached 50% [2].
In recent years, epidemiological data has found that the incidence of IBD has increased year by year, and the main affected population is young adults.
The treatment of the disease is patients and society.
Both bring serious burdens
.
Common complications of CD (1) Gastrointestinal bleeding (2) Intestinal stenosis and obstruction (3) Intestinal perforation (4) Abdominal abscess (5) Anal fissure, anal fistula and perianal abscess (6) Carcinogenesis (7) Extraintestinal complications CD treatment drugs and program treatment drugs: mesalazine, hormones, immunosuppressants, biological agents, etc.
[3]
.
Although traditional therapeutic drugs can improve symptoms, they cannot prevent the underlying inflammatory process.
The emergence of biological agents has greatly changed the treatment methods and treatment goals of IBD, and can induce clinical remission of patients, achieve intestinal mucosal healing, and reduce the rate of surgery and hospitalization.
.
The study in Figure 1 believes that for patients newly suffering from CD, early active use of de-escalation therapy can achieve better clinical efficacy than traditional drug therapy [4]
.
Biologics treatment of CD Infliximab (IFX) was approved for CD indications in China in 2007, officially opening a new era of anti-tumor necrosis factor (TNF) treatment for inflammatory bowel disease in China, bringing new opportunities to patients and doctors Hope
.
In 2020, with the approval of Adalimumab, Verdrizumab, and Usnumab, CD patients have more choices
.
Figure 2 CD therapy in the new era of interleukins.
Usnuzumab is the world's first fully human "dual-targeting" IL-12 and IL-23 inhibitor.
It was approved by the U.
S.
Food and Drug Administration (FDA) as early as 2016.
For the treatment of active CD, the CD indication was approved in China in March 2020
.
Global Phase III clinical studies (UNITI-1, UNITI-2, and IM-UNITI) have shown that the treatment of CD with ulinumumab can quickly improve symptoms and maintain remission, reduce the rate of hospitalization and surgery, and change the course of the disease [5 ]
.
The arrival of Usnumab also symbolizes that IBD treatment has officially entered a new era of interleukin
.
Usinumab is a fully human monoclonal antibody prepared using transgenic technology.
Compared with IFX (chimeric) and adalimumab (phage display technology), it has a lower immunogenicity and anti-drug antibody production rate.
Numab anti-drug antibody production rate <3%
.
2021 DDW announced a comprehensive analysis of the long-term safety data set of Uselnumumab in the treatment of IBD.
The results of the data set analysis showed that the incidence of adverse events in the Uselnumumab group and the placebo group was similar, and the value was higher than that of the Uselnumumab group.
The placebo group had fewer adverse reactions in the treatment of CD.
The results of this analysis showed that the safety of Uselnumumab was good
.
During the induction period of ulinumumab, intravenous induction therapy is performed once, and the maintenance period is subcutaneously injected every 12 weeks (every 8 weeks if necessary)
.
Only 4 subcutaneous injections are required to maintain the number of injections throughout the year.
It is a very convenient biological preparation in the field of CD treatment
.
Figure 3 The 11th Guangzhou International Digestion Summit Forum (GIGIS) and the 9th Asian Inflammatory Bowel Disease Conference and China Inflammatory Bowel Disease Annual Conference will be held in Guangzhou, China on October 14-16, 2021.
There are domestic The most cutting-edge academic information from abroad; scan the QR code below to enter a new era of CD therapy intermediary with us! Scan the QR code below for more information.
References: 1.
Lonberg N.
Nat Biotechnol 2005;23:1117–252.
Peyrin-Biroulet L, Harmsen WS, Tremaine WJ, Zinsmeister AR, Sandborn WJ, Loftus EV.
Surgery in a population-based cohort of Crohn's disease from Olmsted County, Minnesota (1970-2004).
Am J Gastroenterol 2012;107:1693–701.
3.
Consensus opinions on the diagnosis and treatment of inflammatory bowel disease (2018 Beijing) 4.
Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn's disease: an open randomised trial .
Lancet, 20085.
Feagan BG, Sandborn WJ, Gasink C, et al.
Ustekinumab as Induction and Maintenance Therapy for Crohn's Disease.
N Engl J Med 2016;375:1946 -60.
Statement: This content is only for Chinese medical and health professionals, and aims to provide scientific information to and only to medical and health professionals for personal learning and reference purposes
.
If you are not a medical and health professional, please do not participate or spread
.
-End-This article is only used to provide scientific information to medical professionals and does not represent the platform's position
.
The 9th Asian Inflammatory Bowel Disease Conference and China Inflammatory Bowel Disease Annual Conference (AOCC) will be held on October 14-16, 2021 in Guangzhou, China
.
Crohn's disease (CD) is a type of inflammatory bowel disease (IBD), which can occur in any part of the gastrointestinal tract.
It is a chronic, recurrent and non-specific gastrointestinal disease.
The cause of transmural inflammation is unclear so far [1]
.
Half of the patients required surgery within the first 10 years and the risk of postoperative recurrence reached 50% [2].
In recent years, epidemiological data has found that the incidence of IBD has increased year by year, and the main affected population is young adults.
The treatment of the disease is patients and society.
Both bring serious burdens
.
Common complications of CD (1) Gastrointestinal bleeding (2) Intestinal stenosis and obstruction (3) Intestinal perforation (4) Abdominal abscess (5) Anal fissure, anal fistula and perianal abscess (6) Carcinogenesis (7) Extraintestinal complications CD treatment drugs and program treatment drugs: mesalazine, hormones, immunosuppressants, biological agents, etc.
[3]
.
Although traditional therapeutic drugs can improve symptoms, they cannot prevent the underlying inflammatory process.
The emergence of biological agents has greatly changed the treatment methods and treatment goals of IBD, and can induce clinical remission of patients, achieve intestinal mucosal healing, and reduce the rate of surgery and hospitalization.
.
The study in Figure 1 believes that for patients newly suffering from CD, early active use of de-escalation therapy can achieve better clinical efficacy than traditional drug therapy [4]
.
Biologics treatment of CD Infliximab (IFX) was approved for CD indications in China in 2007, officially opening a new era of anti-tumor necrosis factor (TNF) treatment for inflammatory bowel disease in China, bringing new opportunities to patients and doctors Hope
.
In 2020, with the approval of Adalimumab, Verdrizumab, and Usnumab, CD patients have more choices
.
Figure 2 CD therapy in the new era of interleukins.
Usnuzumab is the world's first fully human "dual-targeting" IL-12 and IL-23 inhibitor.
It was approved by the U.
S.
Food and Drug Administration (FDA) as early as 2016.
For the treatment of active CD, the CD indication was approved in China in March 2020
.
Global Phase III clinical studies (UNITI-1, UNITI-2, and IM-UNITI) have shown that the treatment of CD with ulinumumab can quickly improve symptoms and maintain remission, reduce the rate of hospitalization and surgery, and change the course of the disease [5 ]
.
The arrival of Usnumab also symbolizes that IBD treatment has officially entered a new era of interleukin
.
Usinumab is a fully human monoclonal antibody prepared using transgenic technology.
Compared with IFX (chimeric) and adalimumab (phage display technology), it has a lower immunogenicity and anti-drug antibody production rate.
Numab anti-drug antibody production rate <3%
.
2021 DDW announced a comprehensive analysis of the long-term safety data set of Uselnumumab in the treatment of IBD.
The results of the data set analysis showed that the incidence of adverse events in the Uselnumumab group and the placebo group was similar, and the value was higher than that of the Uselnumumab group.
The placebo group had fewer adverse reactions in the treatment of CD.
The results of this analysis showed that the safety of Uselnumumab was good
.
During the induction period of ulinumumab, intravenous induction therapy is performed once, and the maintenance period is subcutaneously injected every 12 weeks (every 8 weeks if necessary)
.
Only 4 subcutaneous injections are required to maintain the number of injections throughout the year.
It is a very convenient biological preparation in the field of CD treatment
.
Figure 3 The 11th Guangzhou International Digestion Summit Forum (GIGIS) and the 9th Asian Inflammatory Bowel Disease Conference and China Inflammatory Bowel Disease Annual Conference will be held in Guangzhou, China on October 14-16, 2021.
There are domestic The most cutting-edge academic information from abroad; scan the QR code below to enter a new era of CD therapy intermediary with us! Scan the QR code below for more information.
References: 1.
Lonberg N.
Nat Biotechnol 2005;23:1117–252.
Peyrin-Biroulet L, Harmsen WS, Tremaine WJ, Zinsmeister AR, Sandborn WJ, Loftus EV.
Surgery in a population-based cohort of Crohn's disease from Olmsted County, Minnesota (1970-2004).
Am J Gastroenterol 2012;107:1693–701.
3.
Consensus opinions on the diagnosis and treatment of inflammatory bowel disease (2018 Beijing) 4.
Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn's disease: an open randomised trial .
Lancet, 20085.
Feagan BG, Sandborn WJ, Gasink C, et al.
Ustekinumab as Induction and Maintenance Therapy for Crohn's Disease.
N Engl J Med 2016;375:1946 -60.
Statement: This content is only for Chinese medical and health professionals, and aims to provide scientific information to and only to medical and health professionals for personal learning and reference purposes
.
If you are not a medical and health professional, please do not participate or spread
.
-End-This article is only used to provide scientific information to medical professionals and does not represent the platform's position
.